Calculate the potential for each of the voltaic cells in Exercise 18.44 when the concentrations of the
Question:
Calculate the potential for each of the voltaic cells in Exercise 18.44 when the concentrations of the soluble species and gas pressures are as follows:![(a) [Fe+] = 0.050 M, [Zn+] = 1.0 10- M (b) [Fe+] = 0.20 M, [Fe+] = 0.010 M, [CI-] = 4.0 X 10-3 M (c) [Br] =](https://dsd5zvtm8ll6.cloudfront.net/images/question_images/1704/7/1/1/350659bd4b6b819a1704711350729.jpg)
Exercise 18.44
For each of the reactions, calculate E° from the table of standard potentials, and state whether the reaction is spontaneous as written or spontaneous in the reverse direction under standard conditions.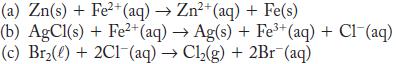
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Related Book For 

Chemistry Principles And Practice
ISBN: 9780534420123
3rd Edition
Authors: Daniel L. Reger, Scott R. Goode, David W. Ball
Question Posted:





