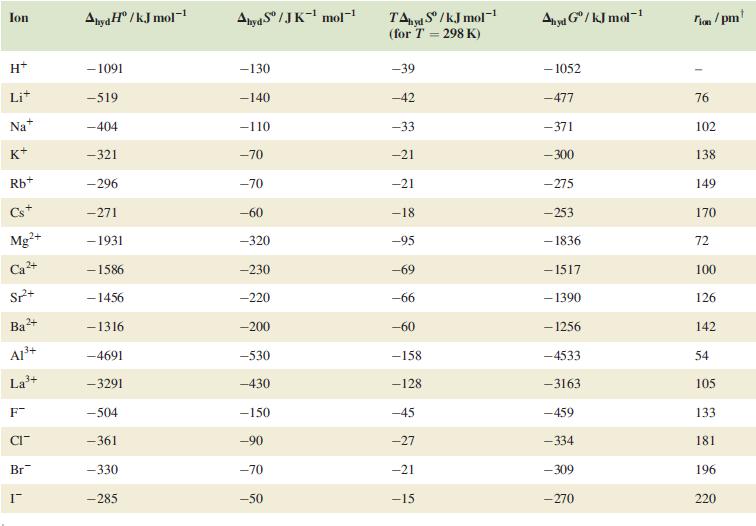
By constructing thermodynamic cycles analogous to those shown in Fig. 8.5, discuss the factors that contribute to
Question:
By constructing thermodynamic cycles analogous to those shown in Fig. 8.5, discuss the factors that contribute to the trend in values of Eo for the group 1 metals Li to Cs.
Figure 8.5

Table 7.7
Transcribed Image Text:
Step (1) Dehydration of the metal ion M²+ (aq) + zeM(s) M² (g) Step (2) -ΣΙΕ Step (3) M(g) Step (1) Dehydration of the hydrogen ion H¹(aq) + ¹/2H₂(g) H*(g) Step (2) -IE₁ Step (3) 1 H(g)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (3 reviews)
To discuss the factors contributing to the trend in standard reduction potentials E for the Group 1 metals Li to Cs we can construct thermodynamic cycles analogous to those shown in Fig 85 These therm...View the full answer

Answered By

Diana Muriuki
As an online math tutor, I have several years of hands-on experience working with students of all ages and skill levels. I hold a Bachelor's degree in Mathematics and a Master's degree in Education. Additionally, I have completed multiple training courses in online teaching and tutoring methods.
Throughout my career, I have worked with students in both individual and group settings, including classroom teaching, after-school tutoring, and online instruction. I am proficient in teaching a wide range of math topics, from basic arithmetic to advanced calculus and statistics.
One of my greatest strengths as a tutor is my ability to adapt my teaching style to meet the unique needs and learning styles of each individual student. I understand that every student is different, and I strive to create a comfortable and supportive learning environment that encourages growth and development.
In addition to my formal education and tutoring experience, I am also a lifelong learner with a passion for mathematics. I am constantly seeking out new resources and methods to improve my own knowledge and skills, and I believe this passion and enthusiasm helps to inspire my students as well.
Overall, my hands-on experience and proficiency as a math tutor are grounded in a combination of formal education, practical experience, and a genuine love of mathematics. I am confident in my ability to help students achieve their goals and succeed in math, and I look forward to the opportunity to work with new students and continue to grow as an educator.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Discuss the factors that make supplier selection and evaluation difficult.
-
Discuss the factors that should be taken into consideration when choosing a depreciation method.
-
Discuss the factors that influence internal pay structures. Based on your own experience, which ones do you think are the most important? Why?
-
How could not having a strategy hurt a brand on social media? What are some content ideas for those who are at the "Awareness" stage of the sales funnel? What does it mean to A/B test on a social...
-
What are some criteria managers should consider to avoid pre-and postacquisition problems?
-
1. It has been suggested by Jamie Lee and Ross professional investment counselor to perform a financial check-up as the first step in investing in mutual funds, even though they are investing $50,000...
-
Nereus Montemayor was an employee of VZ Hogs, a company that raises hogs and produces hog feed. VZ Hogs used an extruder manufactured by Sebright Products, Inc. to create hog feed out of discarded...
-
Comfi Airways, Inc., a small two-plane passenger airline, has asked for your assistance in some basic analysis of its operations. Both planes seat 10 passengers each, and they fly commuters from...
-
Explain how the Pakistan crisis will affect the following companies: a) MEIR Commodities India Pvt. Ltd., an agri commodity trading house exporting sugar, molasses, organics, etc. b) Honda, Toyota,...
-
The following potential diagram is part of that illustrating the redox chemistry of chlorine in aqueous solution at pH0. (a) Calculate the value of E for the reduction of [ClO 3 ] to HClO 2 . (b)...
-
Using data from Appendix 11, and the value for the standard Gibbs energy of formation for PbS of 99 kJ mol 1 , determine a value for K sp for this salt. Data from Appendix 11 The concentration of...
-
What do you understand the principal differences between equality and discrimination to be? Provide examples in support of your understanding.
-
If Berkshire Hathaways A shares are trading at $80,000, what split ratio would it need to bring its stock price down to $40?
-
On January 20, Metropolitan, Inc., sold 10 million shares of stock in an SEO. The market price of Metropolitan at the time was $42.50 per share. Of the 10 million shares sold, 5 million shares were...
-
Which of the following one-year, $1000 bank loans offers the lowest effective annual rate? a. A loan with an APR of 6.1%, compounded monthly b. A loan with an APR of 6.1%, compounded annually, with a...
-
With its current leverage, Impi Corporation will have net income next year of $9.5 million. If Impis corporate tax rate is 30% and it pays 8% interest on its debt, how much additional debt can Impi...
-
It is May 16, 2019, and you have decided to purchase 10 June call contracts on Amazons stock with an exercise price of $1905. Because you are buying, you must pay the ask price. How much money will...
-
Explain how the statement of financial position and statement of income would compare if a company used FIFO instead of weighted-average when the prices of inventory were rising. How would this...
-
Carlton Stokes owns and operates a car-detailing business named SuperShine & Detailing. For $150, Carltons business will hand wash and wax customers cars, vacuum the interior, and thoroughly clean...
-
Describe the difference between a physical vapour and a chemical vapour with respect to the type and stability of the vapour species.
-
Reaction of ZrS 2 (c lattice parameter 583 pm) with [Co( 5 -C 5 H 5 ) 2 ] gives a compound with a c lattice parameter of 1164 pm, and reaction with [Co( 5 -C 5 Me 5 ) 2 ] gives a product with a...
-
(a) Discuss the difference between homogeneous and heterogeneous nucleation from the vapour phase. (b) Which type of nucleation is preferred for the growth of a thin film in this process? (c) Which...
-
how a Christian worldview perspective on personal debt may conflict with how a multinational company leverages debt to finance its operations and growth.
-
Simplify 162 to the form ab.
-
At closing, the earnest money left on deposit with a real estate broker is (1pts) Question 7 - At closing, the earnest money left on deposit with a real estate broker is a credit to the seller. a...

Study smarter with the SolutionInn App


