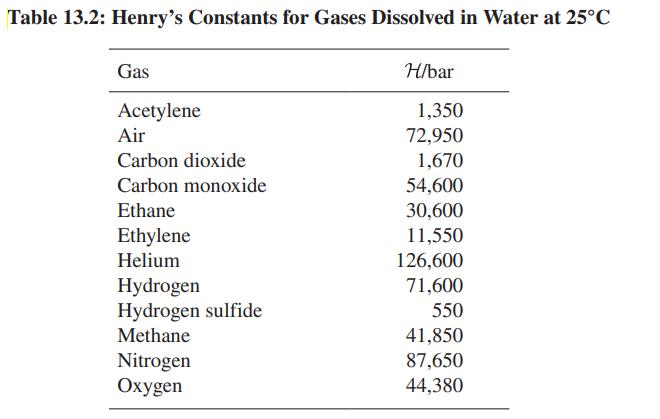
Helium-laced gases are used as breathing media for deep-sea divers. Why? Table 13.2 may provide useful data.
Question:
Helium-laced gases are used as breathing media for deep-sea divers. Why? Table 13.2 may provide useful data.
Transcribed Image Text:
Table 13.2: Henry's Constants for Gases Dissolved in Water at 25°C Gas Acetylene Air Carbon dioxide Carbon monoxide Ethane Ethylene Helium Hydrogen Hydrogen sulfide Methane Nitrogen Oxygen H/bar 1,350 72,950 1,670 54,600 30,600 11,550 126,600 71,600 550 41,850 87,650 44,380
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 77% (9 reviews)
Heliumlaced gases are used as breathing media for deepsea divers for several important reasons primarily related to its low solubility in blood and ti...View the full answer

Answered By

Abdul Wahab Qaiser
Before working at Mariakani, I volunteered at a local community center, where I tutored students from diverse backgrounds. I helped them improve their academic performance and develop self-esteem and confidence. I used creative teaching methods, such as role-playing and group discussions, to make the learning experience more engaging and enjoyable.
In addition, I have conducted workshops and training sessions for educators and mental health professionals on various topics related to counseling and psychology. I have presented research papers at conferences and published articles in academic journals.
Overall, I am passionate about sharing my knowledge and helping others achieve their goals. I believe that tutoring is an excellent way to make a positive impact on people's lives, and I am committed to providing high-quality, personalized instruction to my students.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Introduction To Chemical Engineering Thermodynamics
ISBN: 9781260597684
9th International Edition
Authors: J.M. Smith, Mark Swihart Hendrick C. Van Ness, Michael Abbott
Question Posted:
Students also viewed these Engineering questions
-
Planning is one of the most important management functions in any business. A front office managers first step in planning should involve determine the departments goals. Planning also includes...
-
On June 23, 2018, in Thailand, a group of 12 boys aged between 11 and 17 from the local football team, named the Wild Boars, and their 23-year-old assistant coach entered the Tham Luang cave. Tham...
-
Write a literature review for your study. See below for an example of a literature review. Your literature review should provide both analysis and synthesis of previous studies as related to the...
-
Attijari Wafa Bank distributed on May 2022 a dividend per share of 5 MAD / share. The company is planning to have next 3 years a growth rate of 5% then after 7%. If the required rate of return is 12%...
-
Assume the correlation coefficient between Baker Fund and the S&P 500 Stock Index is .70. What percentage of Baker Funds total risk is specific (i.e., nonsystematic)?
-
The least-squares regression equation Å· = 15.2 + 10.7t has been fitted to the shipment data in the table. Calculate the Durbin-Watson d statistic and test for positive autocorrelation...
-
Analyze the chemical process data in Table B. 5 for evidence of multicollinearity. Use the variance inflation factors and the condition number of \(\mathbf{X}^{\prime} \mathbf{X}\). Run No. y X x2 X3...
-
A business executive says, The financial perspective of the balanced scorecard indicates how the organization adds value to shareholders. I am involved with two organizations: a small business that...
-
Beginning raw materials inventory $96,000 Beginning work-in-process inventory 168,000 Beginning finished goods inventory 600,000 Raw materials purchases 684,000 Wages paid 372,000 Applied...
-
Derive the stiffness matrix for a beam element with a nodal linkagethat is, the shear is 0 at node i, but the usual shear and moment resistance are present at node j (see following figure). m; m, L....
-
A system formed initially of 2 mol CO 2 , 5 mol H 2 , and 1 mol CO undergoes the reactions: Develop expressions for the mole fractions of the reacting species as functions of the reaction coordinates...
-
Which is the more effective way to increase the coefficient of performance of a Carnot refrigerator: to increase T C with T H constant, or to decrease T H with T C constant? For a real refrigerator,...
-
Use of special dress, manners, gestures, and vocabulary words when meeting a prospective employer in a job interview are all examples of how people use ____________ in daily life. (a) projection (b)...
-
What are the different types of advances of a bank according to RBI prudential norms?
-
You are the auditors of Apollo Services Ltd. and you have recently been reading a report on a similar company that criticised the auditors for failing to comment on the existence of the material...
-
What do you mean by Auditors Report?
-
Write short notes on compulsory tax audit.
-
What special points the auditor has to consider in conducting audit in the following institutions: (a) Charitable institution. (b) Educational institution.
-
Burns Boats wants to assemble 50 boats per 8-hour day, using a production line. Total task time for each boat is 45 minutes. a. What is the takt time? What is the theoretical mini mum number of...
-
One of the significant and relevant accounts for this cycle is equipment. For this account, what would typically be the most relevant assertions for the auditor to consider? Why is it important for...
-
A preliminary evaluation of a new process concept has produced a waste stream of the composition given below. It is desired to reduce the waste stream to 10% of its original mass while recovering...
-
According to Gmehling et al. (1994),23 the system acetone + water shows azeotropes at: (1) 2793 mmHg, 95.1 mol% acetone, and 100C; and (2) 5155 mmHg, 88.4 mol% acetone and 124C. What azeotropic...
-
Activity coefficients are an implicit part of the equation of state but they can be determined explicitly by comparing the definitions of the K-ratios. Using the k ij value fit at x e = 0.415,...
-
Silver Company makes a product that is very popular as a Mother s Day gift. Thus, peak sales occur in May of each year, as shown in the company s sales budget for the second quarter given below:...
-
! 10 Required information Problem 9-7B Calculate the issue price of a bond and prepare amortization schedules (LO9-5, 9-7) [The following information applies to the questions displayed below.]...
-
Exercise 13-6 (Algo) Prepare a Statement of Cash Flows; Free Cash Flow [LO13-1, LO13-2, LO13-3] Comparative financial statement data for Carmono Company follow: Assets Cash Accounts receivable...

Study smarter with the SolutionInn App


