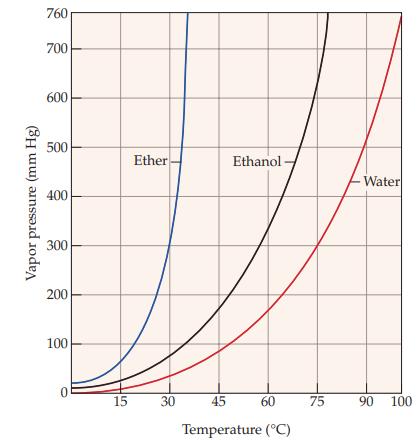
Refer to Figure 11.5 and determine the approximate boiling point of ether. Figure 11.5 Vapor pressure (mm
Question:
Refer to Figure 11.5 and determine the approximate boiling point of ether.
Figure 11.5
Transcribed Image Text:
Vapor pressure (mm Hg) 760 700 600 500 400 300 200 100 0 15 Ether 30 Ethanol 45 60 Temperature (°C) 75 > -Water 90 100
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
The normal boiling point of a liquid is the temperature at which th...View the full answer

Answered By

OTIENO OBADO
I have a vast experience in teaching, mentoring and tutoring. I handle student concerns diligently and my academic background is undeniably aesthetic
4.30+
3+ Reviews
10+ Question Solved
Related Book For 

Introductory Chemistry Concepts And Critical Thinking
ISBN: 9780321804907
7th Edition
Authors: Charles Corwin
Question Posted:
Students also viewed these Sciences questions
-
Refer to Figure 11.5 and determine the approximate boiling point of ethanol. Figure 11.5 Vapor pressure (mm Hg) 760 700 600 500 400 300 200 100 15 Ether 30 Ethanol 45 60 Temperature (C) 75 Water 90...
-
The warranty and customer service that come with the purchase of a car, are part of its _______product. Multiple Choice durable branded core actual augmented
-
Predict the approximate boiling point of (a) Ethylbenzene (b) Propylbenzene (c) P-xylene
-
A group of fraud examiners is coordinating an investigation at a local law firm. Several lawyers at the firm are suspected of overbilling clients, possibly creating fake client accounts, and then...
-
Nichols Corporations value of operations is equal to $500 million after a recapitalization (the firm had no debt before the recap). It raised $200 million in new debt and used this to buy back stock....
-
(CPA, adapted) The Scarborough Corporation manufactures and sells two products: Thingone and Thingtwo. In July 2011, Scarboroughs budget department gathered the following data to prepare budgets for...
-
An electron moves in a straight line at a speed of \(6.0 \times 10^{7} \mathrm{~m} / \mathrm{s}\). Calculate the magnitude and direction of the magnetic field at a position \(5.0 \mathrm{~mm}\)...
-
Dr. Mark Greenberg practices dentistry in Topeka, Kansas. Greenberg tries hard to schedule appointments so that patients do not have to wait beyond their appointment time. His October 20 schedule is...
-
Mr. Mutwiri opened a business in Meru town at the beginning of the year. During the first month of operation the following transactions were completed. 2014. January 1 Deposited sh 20,000 in the bank...
-
Which of the following is an observed property of liquids? (a) Liquids are more dense than gases. (b) Liquids expand and compress significantly. (c) Liquids have a fixed shape and variable volume....
-
Which of the following liquids has the stronger intermolecular attraction between molecules: CH 3 CH 2 OH or CH 3 OCH 3 ?
-
How does a high-pitch musical note relate to frequency?
-
Calculate the total returns (TR) and the return relatives (RR) in term of Euro for the following assets: i. A preferred stock bought for 70 per share, held one year during which 5 per dividend are...
-
What contribution would make and to whom if better understanding the investor behavior during covid 19?
-
(b) A recursive algorithm has been developed as shown below: Input: List A = (a,..., ay) such that N 1 Output: value x foo(A) if length(A) == 1 return (a * a) A' = (a,..., aN-1) q = foo(A') return q...
-
The function has a maximum of at x = The function has a minimum of at x = The function is increasing on the interval(s): The function is decreasing on the interval(s): The domain of the function is:...
-
Items Pension Worksheet General Journal Entries Memo Record Annual Pension Pension Asset/ Projected Benefit Plan Expense Cash Liability Obligation Assets $ - $128000 cr $128000 dr Balance, Jan. 1,...
-
Carter wants to retire from his florist business, and his long-time employee, Howard, would like to take over the business. Identify the tax issue(s) posed by the facts presented. Determine the...
-
Under what conditions is the following SQL statement valid?
-
Curtis Bog, chief financial officer of Sphagnum Paper Corporation, is reviewing a consultant?s analysis of Sphagnum?s weighted-average cost of capital. The consultant proposes Mr. Bog wants to check...
-
Nevada Hydro is 40 percent debt-financed and has a weighted-average cost of capital of 9.7 percent: Banker?s Tryst Company is advising Nevada Hydro to issue $75 million of preferred stock at a...
-
Sometimes APV is particularly useful in international capital investment decisions. What kinds of tax or financing side effects are encountered in international projects?
-
Happy, Inc. was founded 9 years ago by siblings Brandon and Rachael Happy. The company manufactures and installs commercial heating, ventilation, and cooling (HVAC) units. Happy, Inc. experienced...
-
Braile Gear Works sells a single gear for a price of $73.00 per unit. The variable costs of the gear are $41.00 per gear and annual fixed costs are $548,800. Required: a. What is the break-even level...
-
A lease agreement that qualifies as a finance lease calls for annual lease payments of $10,000 over a five-year lease term (also the asset's useful life), with the first payment on January 1, the...

Study smarter with the SolutionInn App


