From what epoxide and what nucleophile could each of the following compounds be prepared? (Assume each is
Question:
From what epoxide and what nucleophile could each of the following compounds be prepared? (Assume each is racemic.)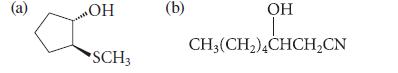
Transcribed Image Text:
(a) ОН SCH3 (b) OH CH3(CH₂)4CHCH₂CN
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
The strategy in this problem is to let the OH group originate from the epo...View the full answer

Answered By

Lisper Wanja
I am an experienced and highly motivated writer with a passion for the skills listed. I have a proven track record of my expertise and my aim is to deliver quality, well-detailed and plagiarism free projects. My genuine passion for writing combined with my ongoing professional development through school and research makes me an ideal candidate within for any assignment.
4.90+
233+ Reviews
388+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
a. How could each of the following compounds be prepared from a hydrocarbon in a single step? 1. 2. 3. b. What other organic compound would be obtained from each synthesis? OH Br Br CH CH HO
-
From what epoxide and what nucleophile colld each of the following compound be prepared Inppued? (Assums each is racemic.) C,H OH/H,O CH CH2 sodium azide
-
From which alkyne could each of the following compounds be prepared by acid- catalyzed hydration? (a) (c) CH3CCHCHCH3 (b) O (CH3)3C-C-CH3
-
1. Describe briefly how computers work and become tools for committing cyber-related crimes. 2. Use of proxies and VPNs is one of the methods cybercriminals try to cover their digital track and...
-
John Verner is the controller for BioMedic, Inc., a biotechnology company. John is finishing his preparation of the preliminary financial statements for a meeting of the board of directors scheduled...
-
Explore the Critical Thinking Community Web site at criticalthinking.org. Identify three important topics currently being discussed, and describe your findings.
-
Go to the St. Louis Federal Reserve FRED database, and find data on real GDP (GDPC1), potential GDP (GDPPOT), and the unemployment rate (UNRATE) from 1960 to the most recent period. For the...
-
The Big Black Bird Company produces fiberglass camper tops. The process for producing the tops must be controlled so as to keep the number of dimples low: When the process was in control, the...
-
An aluminum flagpole is 34 m high. By how much does its length increase as the temperature increases by 23 C? (The linear expansion coefficient of aluminum is 23 x 10-6 /C)
-
Explain each of the following facts with a mechanistic argument. (a) When butyl methyl ether (1-methoxybutane) is treated with HI and heat, the initially formed products are mainly methyl iodide and...
-
The chlorohydrin trans-2-chlorocyclohexanol reacts rapidly in base to form an epoxide. The cis stereoisomer, however, is relatively unreactive and does not give an epoxide. Explain why the two...
-
In many states, a bartender (under so-called dram shop laws), friend, party host, or other person who serves liquor to an already-intoxicated person may be held vicariously liable for any damages...
-
Air at 200F and 1 atm absolute pressure is to be cooled to 70F by passing it through a 2-in. ID copper pipe submerged in an ice bath. The air flow rate is 70 lb/h. Neglecting the heat transfer...
-
Any other improvement can be done on question 3 solution if the number of CPU cores is eight. Question 3 In given K sorted Lists of fixed length M. Also, given a final output list of length M*K. Give...
-
A company has current liabilities of $500 million, and its current ratio is 2.0. What is the total of its current assets? ($1,000 million) If this firms quick ratio is 1.6, how much inventory does it...
-
Which firms are the biggest players in the sector often referred to as the sharing economy or collaborative consumption? Have you used any of these firms? What was your experience like? Would you use...
-
Name traditional firms that have experimented with their own collaborative consumption efforts. Conduct additional research online. Are these efforts successful, failed, or is it too early to tell?...
-
What type of income does a sole proprietor of a business receive?
-
Describe a job you have had in the past or a job you are very familiar with. Indicate the negative aspects of the job and how it could be improved with current human resource management techniques.
-
Assume that acetyl CoA containing a 14C isotopic label in the carboxyl carbon atom is used as starting material for the biosynthesis of mevalonate, as shown in figure. At what positions in mevalonate...
-
Assume that acetyl CoA containing a 14C isotopic label in the carboxyl carbon atom is used as starting material and that the mevalonate pathway is followed. Identify the positions in ?-cadinol where...
-
Assume that acetyl CoA containing a 14C isotopic label in the carboxyl carbon atom is used as starting material and that the mevalonate pathway is followed. Identify the positions in squalene where...
-
If demand of a product for five periods is as follows: Period 1 2 3 4 5 Demand ( Units ) 3 0 3 5 4 0 4 2 4 5 What is the forecast for the 6 th period using a three - period moving average...
-
Consider the below data for inventory items Annual total demand 5000 units Number of working days 20 Lead time 4 Standard division of total demand 20 units Confidence level demands 95 % The value of...
-
Suppose a construction materials company sells gravel for road construction projects. They order gravel periodically from their supplier using a continuous review policy. The average demand is 320...

Study smarter with the SolutionInn App


