In each of the following cases, the labeled protons are constitutionally equivalent. Determine whether the labeled protons
Question:
In each of the following cases, the labeled protons are constitutionally equivalent. Determine whether the labeled protons in each case are expected to have identical or different chemical shifts.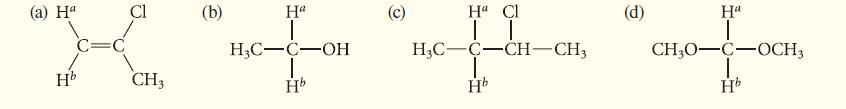
Transcribed Image Text:
(a) Ha Hb Cl CH3 (b) Ha 1 H3C-C-OH Hb O Ha Cl | | H3C-C-CH-CH3 Hb (d) Ha 1 CH3O-C-OCH3 Hb
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
Apply the principles of Sec 109A to determine whether the protons in question are chemically equivalent If the two protons are chemically equivalent t...View the full answer

Answered By

JAPHETH KOGEI
Hi there. I'm here to assist you to score the highest marks on your assignments and homework. My areas of specialisation are:
Auditing, Financial Accounting, Macroeconomics, Monetary-economics, Business-administration, Advanced-accounting, Corporate Finance, Professional-accounting-ethics, Corporate governance, Financial-risk-analysis, Financial-budgeting, Corporate-social-responsibility, Statistics, Business management, logic, Critical thinking,
So, I look forward to helping you solve your academic problem.
I enjoy teaching and tutoring university and high school students. During my free time, I also read books on motivation, leadership, comedy, emotional intelligence, critical thinking, nature, human nature, innovation, persuasion, performance, negotiations, goals, power, time management, wealth, debates, sales, and finance. Additionally, I am a panellist on an FM radio program on Sunday mornings where we discuss current affairs.
I travel three times a year either to the USA, Europe and around Africa.
As a university student in the USA, I enjoyed interacting with people from different cultures and ethnic groups. Together with friends, we travelled widely in the USA and in Europe (UK, France, Denmark, Germany, Turkey, etc).
So, I look forward to tutoring you. I believe that it will be exciting to meet them.
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Planning is one of the most important management functions in any business. A front office managers first step in planning should involve determine the departments goals. Planning also includes...
-
Based on Westlaw's data on the case Christoff v. Nestl USA, INC., I did the brief case. My question is, is there any need to add and adjust the content and form of the case brief, especially the...
-
Specify whether the labeled protons in each of the following structures would be expected to have the same or different chemical shifts. (a) (b) CH3 H,C CH3 HC C-CH CI Hb
-
You are given the following as it relates to the price of a pound of flour in Adam Island, 2016 to 2022: 2016-$1.35 2017 - $1.50 2018 - $1.65 2019 - $1.85 2020 - $2.15 2021 - $2.00 2022 - $2.10 Using...
-
What effect does a debit memo in a bank statement have on the Cash account? What effect does a credit memo in a bank statement have on the Cash account?
-
Indicate whether you think the following claims regarding takeovers are true or false. In each case, provide a brief explanation for your answer. a. By merging competitors, takeovers have created...
-
In February 2014, defendant Ibrahim M. Shihadeh, d/b/a Creative Designs Kitchen and Baths, agreed to purchase 25% of his anticipated natural gas needs at a fixed price for the 201415 and 201516...
-
Value engineering, target pricing, and locked-in costs. Pacific Dcor, Inc., designs, manufactures, and sells contemporary wood furniture. Ling Li is a furniture designer for Pacific. Li has spent...
-
First Draw an ER Model please and then covert it into relational model seperate figures required thank you Each cinema is identified by its name and has its residency at an address which consists of...
-
Determine the structure of the compound with the formula C 6 H 13 Cl that has the NMR spectrum shown in Fig. 13.13. FIGURE 13.13 The NMR spectrum for Study Problem 13.4. 2400 absorption 8 2100 7 1800...
-
A compound contains carbon, hydrogen, oxygen, and one nitrogen. Classify each of the following fragment ions derived from this compound as an odd-electron or an even-electron ion. Explain. (a) The...
-
Solve each problem The weight of an object varies inversely as the square of its distance from the center of Earth. If an object 8000 mi from the center of Earth weighs 90 lb, find its weight when it...
-
Dr. Doe firmly believes in the terminally ills right to die. He has contracts to assist Richard Roe, an 80-year-old man dying of cancer, in committing suicide. The fee agreed on is $1,000. After Mr....
-
Search online to read about Jeff Bezoss management of his new executives. Specifically, explore Amazon.coms S-Team meetings (management.fortune.cnn.com/2012/11/16/jeff-bezos-amazon/). Why does Bezos...
-
An increase in _________ GDP guarantees that more goods and services are being produced by an economy. a. nominal b. real
-
Consider there is a loop in a linked list, Write a program to remove loop if there is a loop in this linked list. //Sorts a given list by selection sort //Input: An array A[0..n-1] of orderable...
-
When have you heard or said I have no other choice?
-
Merlene owns a bookstore. The store needs repainting, but she is short of cash to hire a painter. Fred is a painter who enjoys fine mystery novels. Merlene makes a deal with Fred to have him paint...
-
When you weigh yourself on good old terra firma (solid ground), your weight is 142 lb. In an elevator your apparent weight is 121 lb. What are the direction and magnitude of the elevator's...
-
There are four resonance structures for anthracene, one of which is shown. Draw the otherthree. Anthracene
-
There are five resonance structures of Phenanthrene, one of which is shown. Draw the otherfour. Phenanthrene
-
Look at the five resonance structures for Phenanthrene (Problem 15.26) and predict which of its carboncarbon bonds is shortest. Phenanthrene
-
An investor purchased a stock for $33.62 and sold it one year later for $34.86. The investor also received a dividend payment of $1.47. What was the investor's realized dividend yield? (Enter your...
-
1. Show the output of the following program: #include using namespace std; int mystery (int); int main() { } int n; for (n = 1; n
-
For Selling and Administrative Expense Budget do you incorporate budgeted sales numbers or production numbers? Why or why not ?Explain

Study smarter with the SolutionInn App


