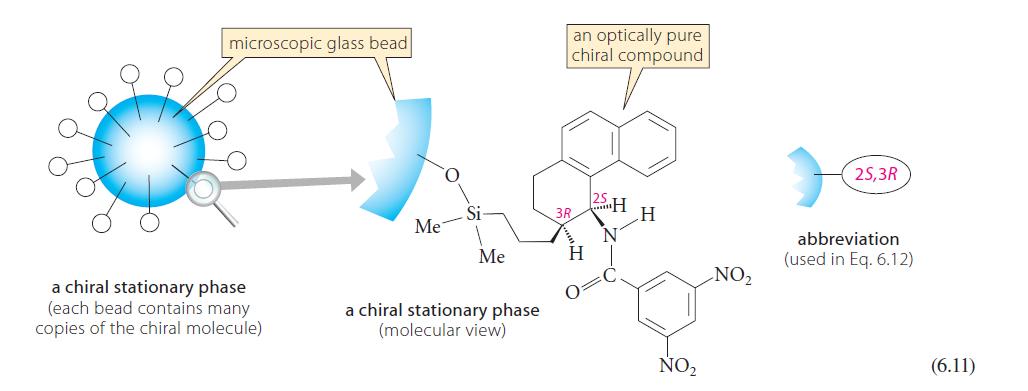
The enantiomeric resolution in Fig. 6.16 used the chiral stationary phase (CSP) in Eq. 6.11. How would
Question:
The enantiomeric resolution in Fig. 6.16 used the chiral stationary phase (CSP) in Eq. 6.11. How would the enantiomeric resolution in Fig. 6.16 be affected if
(a) The enantiomer of the CSP in Eq. 6.11 were used?
(b) The racemate of the CSP in Eq. 6.11 were used?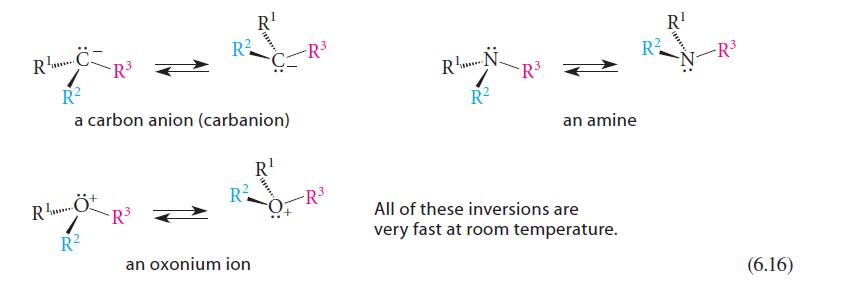
Transcribed Image Text:
CR²³ 3+ R... R² a carbon anion (carbanion) RÖ R² R² -R³ R² R¹ an oxonium ion R¹ -R³ RN R² -R³ R¹ RN-R³² an amine All of these inversions are very fast at room temperature. (6.16)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (3 reviews)
a If the enantiomer of the CSP were used the elution ...View the full answer

Answered By

Ali Khawaja
my expertise are as follows: financial accounting : - journal entries - financial statements including balance sheet, profit & loss account, cash flow statement & statement of changes in equity -consolidated statement of financial position. -ratio analysis -depreciation methods -accounting concepts -understanding and application of all international financial reporting standards (ifrs) -international accounting standards (ias) -etc business analysis : -business strategy -strategic choices -business processes -e-business -e-marketing -project management -finance -hrm financial management : -project appraisal -capital budgeting -net present value (npv) -internal rate of return (irr) -net present value(npv) -payback period -strategic position -strategic choices -information technology -project management -finance -human resource management auditing: -internal audit -external audit -substantive procedures -analytic procedures -designing and assessment of internal controls -developing the flow charts & data flow diagrams -audit reports -engagement letter -materiality economics: -micro -macro -game theory -econometric -mathematical application in economics -empirical macroeconomics -international trade -international political economy -monetary theory and policy -public economics ,business law, and all regarding commerce
4.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The antitumor drug gimatecan is available as nearly pure (S)-enantiomer. Neither pure (R) enantiomer nor a racemic (equal) mixture of the two enantiomers are available. To measure small quantities of...
-
The following additional information is available for the Dr. Ivan and Irene Incisor family from Chapters 1-5. Ivan's grandfather died and left a portfolio of municipal bonds. In 2012, they pay Ivan...
-
How will the total cost of borrowing be affected if a bond is sold (a) At a discount and (b) At a premium?
-
General-equilibrium effects with labor complementarity. Consider an economy comprised of 100 cities. Each city initially contains 1 million each of high school dropouts, high school graduates,...
-
The Budgetary Comparison Schedule for the City of Salem appears in Illustration 216. Assume the general and subsidiary ledgers for the General Fund were lost after a water pipe burst. You are charged...
-
What are the basic problems arising in international trade financing, and how do the main financing instruments help solve those problems?
-
Compare and contrast the ethical approaches of its legal, therefore, its ok and the ends justify the means. Are there similarities? Are there differences?
-
E-mail messages sent over the Internet are broken up into electronic packets that may take a variety of different paths to reach their destination where the original message is reassembled. Suppose...
-
Imagine that you lend $5,000 to a friend at 7%, and say, "Pay me back when you get a job." Five years later, your friend gets a job and pays you back. Your friend assumed that you meant simple...
-
The difference in the standard free energies of formation for 1-butene and 2-methylpropene is 13.4 kJ mol 1 (3.2 kcal mol 1 ). (See the previous problem for a definition of G f .) (a) Which compound...
-
Use the principles of Sec. 1.3B to predict the geometry of BF 3 . What hybridization of boron is suggested by this geometry? Draw an orbital diagram for hybridized boron similar to that for the...
-
If sufficient data are provided, complete the blank cells in the following table of properties of water. In the last column describe the condition of water as compressed liquid, saturated mixture,...
-
Rajasthani Umbrella is a seasonal business that sells umbrellas. At the peak of its rainy selling season, the firm has INR 2,500,000 in cash, INR 3,600,000 in inventory, INR 400,000 in accounts...
-
Derwent Ltd. has announced that the earnings before income and taxes is going to be 300,000 for the current year. Assuming corporate tax rate for Derwent Ltd. is a flat 30%, compute the firms profit...
-
Jennifer has invested in two schemes. The first scheme has a required return of 12% and will produce a stream of 300 at the end of each year indefinitely. The second scheme has a required return of...
-
Ocean Terminal Company Ltd. started its business in 2019 with retained earnings of $68.45 million. It paid two semiannual dividends of $0.23 per share to 3.25 million preferred stockholders during...
-
Suppose you have been offered an investment opportunity that will pay you $500 at the end of every year, starting one year from now and continuing forever. Assume the relevant discount rate is 6%. a....
-
Identify costs that a chemical plant may incur after 20 years of operation (in connection with a planned closing of the plant).
-
What is removed during each of the three stages of wastewater treatment: primary, secondary, and tertiary? During which state would you expect items to be recovered that were accidentally flushed,...
-
How can you account for the fact that 2, 2, 6-trimethylcyclohexanone yields no detectable aldol product even though it has an acidic hydrogen?
-
Cinnamaldehyde, the aromatic constitution of cinnamon oil, can be synthesized by a mixed aldol condensation. Show the starting materials you would use, and write thereaction. CHO Cinnamaldehyde
-
The bicycle ketone shown below does not undergo aldol self-condensation even though it has two ? hydrogen atoms. Explain.
-
Let c (t) = ei + 9 sin(t)j + tk and c (t) = ei + 2 cos(t)j - 10tk. Determine the components of the result, a, b, and c, where d [c (t) x c (t)] = (a, b, c) (Express numbers in exact form. Use...
-
Let f X- Y be an onto function, and let B be a subset of Y. Show that f(f(B)) = B. Give an example showing that equality need not hold if f is not and onto function.
-
(1 point) Given the function f(x)= (in blue), consider the functions g (in green) and h (in red) graphed below which are continuous on (0, c Assuming the graphs continue in the same way as x goes to...

Study smarter with the SolutionInn App


