Propose a plausible synthesis for the following transformation. Enantiomer
Question:
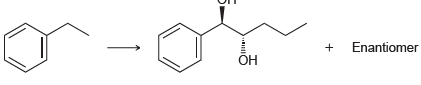
Transcribed Image Text:
Enantiomer ОН
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 38% (13 reviews)
1 Br2 hv 2 NaOEt ...View the full answer

Answered By

Daniel Kimutai
I am a competent academic expert who delivers excellent writing content from various subjects that pertain to academics. It includes Electronics engineering, History, Economics, Government, Management, IT, Religion, English, Psychology, Sociology, among others. By using Grammarly and Turnitin tools, I make sure that the writing content is original and delivered in time. For seven years, I have worked as a freelance writer, and many scholars have achieved their career dreams through my assistance.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Propose a plausible synthesis for each of the following transformations: a. b. c. d. e. f. g. h. Br Br
-
Propose a plausible synthesis for each of the following transformations: a. b. c. Br Br
-
Propose a plausible synthesis for each of the following transformations: a. b. c. d. e. f. g. Br Br
-
LOCATE APPROPRIATE CPT CODES ICD-10-CM (CPT) FOR PROCEDURES BELOW- Outpatient procedures only 1. INCISION AND DRAINAGE OF A CYST --- 2. DEBRIDEMENT - 3. SIMPLE REPAIR OF A SUPERFICIAL WOUND- 4....
-
Repeat Exercise 3.11, maintaining the singly linked list in sorted order. Assume that a singly linked list is implemented with a header node, but no tail node, and that it maintains only a reference...
-
Birth weights in the United States are normally distributed with a mean of 3420 g and a standard deviation of 495 g. a. What percentage of babies is born with a weight greater than 3500 g? b. Find...
-
Consider the hypothetical perfect separation of a mixture of ethylene and ethane into pure products by distillation as shown in Figure 10.39. Two schemes are to be considered: conventional...
-
A bicycle manufacturer currently produces 300,000 units a year and expects output levels to remain steady in the future. It buys chains from an outside supplier at a price of $2 a chain. The plant...
-
Discuss the principles of structured catalysts and reactors in process intensification, analyzing the design and performance characteristics of structured packing, monolithic reactors, and...
-
The balance sheet items for Collier Butcher Shop (arranged in alphabetical order) were as follows at July 1, 2015. (You are to compute the missing figure for Retained Earnings.) During the next few...
-
Draw a Lewis structure for each of the compounds below: a) CH 2 = CHOCH 2 CH(CH 3 ) 2 b) (CH 3 CH 2 ) 2 CHCH 2 CH 2 OH c) (CH 3 CH 2 ) 3 COH d) (CH 3 ) 2 C = CHCH 2 CH 3 e) CH 2 = CHCH 2 OCH 2 CH(CH...
-
Which advantage does a typical developing country have in attempting to draw foreign investment? a. Very low wages b. Poor education c. Easy profit repatriation d. Independent central banks
-
Worried that he had lost the support of his party, the Prime Minister forcefully _______ his controversial statement that healthcare would not be a priority. a. Recanted b. Affirmed c. Validated d....
-
Briefly explain whether each of the following represents a public good. a. The Monaco Grand Prix (formerly the Formula One auto race) held at the Circuit de Monaco b. The samba street parade held...
-
Each instance that follows is an example of one of the four types of market failure discussed in this chapter. In each case, identify the type of market failure and defend your choice briefly. a....
-
New PhDs in economics entering the job market find that academic jobs (jobs teaching at colleges and universities) pay about 30 percent less than nonacademic jobs such as working at a bank or a...
-
In a significant decision regarding patent eligibility, Alice Corp. v. CLS Bank International, the U.S Supreme Court in June 2004 effectively raised the bar for software patents by declining a patent...
-
Assume you are a student in a university town with a population of 200,000. Your parents will be visiting soon and you want to take them out for dinner. Name some of the local and chain restaurants...
-
This insert describes the activities of StellaService, a company that provides online retailers with information about how effective their online activities are in the eyes of consumers. How do the...
-
In Exercises delete part of the domain so that the function that remains is one-to-one. Find the inverse function of the remaining function and give the domain of the inverse function. f(x) = 16x4 -3...
-
Predict the major product(s) of the followingreactions: (b) (a) CH3CH2COCI AICI3 CH3CH2CI AICI3 (c) (d) N(CH2CH3}2 CO2H HNO3 so3 H2SO4 H2SO,
-
Aromatic iodination can be carried out with a number of reagents, including iodine mono chloride, ICI. What is the direction of polarization of ICI? Propose a mechanism for the iodination of an...
-
The sulfonation of an aromatic ring with SO3 and H2SO4 is reversible. That is, heating benzenesulfonic add with H2SO4 yields benzene. Show the mechanism of the de-sulfonation reaction. What is the...
-
Water flows through a pipe section that tapers from an internal diameter of 200 mm down to 100 mm over a distance of 1 m. Determine the pressure drop over the section for a flow of 0.06 m3s-1. The...
-
Calculate the binding energy per nucleon for Be, 59 Co, 208 pb, and 195 pt. (For the atomic masses, see this table. Enter your answers to at least two decimal places.) (a) Be MeV/nucleon 59 Co...
-
A.Final ProjectTeamProjectCustom Schema: 200 points Extra Credit Group of 3 people : Designer and Developer ( ERD, tables, data ) Team Leader/Power User ( Business Questions/Queries, Real Life...

Study smarter with the SolutionInn App


