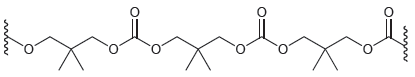
Should this polymer be classified as a chain-growth polymer or a step-growth polymer?
Question:
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (5 reviews)

Answered By

Branice Buyengo Ajevi
I have been teaching for the last 5 years which has strengthened my interaction with students of different level.
4.30+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Polyformaldehyde, sold under the trade name Delrin, is a strong polymer used in the manufacture of many guitar picks. It is prepared via the acid-catalyzed polymerization of formaldehyde. (a) Draw...
-
Consider the structure of the following polymer. (a) Draw the monomers you would use to prepare this polymer. (b) Determine whether this polymer is a step-growth polymer or a chain-growth polymer....
-
The step-growth polymer nylon 6 is prepared from caprolactam. The reaction involves initial reaction of caprolactam with water to give an intermediate open?chain amino acid, followed by healing to...
-
You are an audit supervisor of PricewaterhouseCoopers (PwC) and are planning the audit of your client, Blister Pharmaceuticals co. which manufactures and imports sanitary and cleaning products...
-
Gourmet Bones manufactures its own brand of pet chew bones. At the end of December 2018, the accounting records showed the following: Other information: Direct materials purchases...
-
A program to decrease littering was carried out in four cities in California's Central Valley starting in August 2011. The amount of litter in the streets (average pounds of litter collected per...
-
A chemical laboratory was facing issues with the concentration of the sulfuric acid they prepared. The first step was to collect data on the magnitude of the problem. Of 5,186 recently supplied acid...
-
Global Chemical Company, located in Buenos Aires, Argentina, recently received an order for a product it does not normally produce. Since the company has excess production capacity, management is...
-
If there is at least one state that does not fit the pattern of having a higher average percentage of returned sales in January than the rest of the year, what would your next diagnostic analysis...
-
You have estimated the following cash flows on a project: Year Cash Flow to Equity ($) 0 .. 5,000,000 1 .. 4,000,000 2 .. 4,000,000 3 .. 3,000,000 Plot the NPV profile for this project. What is the...
-
Polyisobutylene cannot be described as isotactic, syndiotactic, or atactic. Explain.
-
Would these pairs of monomers form chain-growth or step-growth polymers? (a) (b) -NH2 H,N- CI CI CI CN
-
How is the data-driven, six-step detection approach relevant to e-business fraud detection?
-
How welcoming do you feel Americans are of newcomers?What are some of the causes of anti-immigrant sentiment? Is it fair to label Americans as prejudiced against immigrants? Why or why not?
-
Your company is considering whether it should tender for two contracts (MS1 and MS2) on offer from a government department for the supply of certain components. The company has three options: tender...
-
Explain on a broad overview of the competencies a human resource executive needs to become a strategic leader in an organization.
-
true or false:With a zero-rate coupon instrument that matures in five years, the amount of interest paid in the second year is zero.
-
What is the quotient of the division (4x3 + 2x - 7x+2) (x-3)? (Simplify your answer completely.) X
-
What are the characteristics of a public bond? (Think in terms of comparing it to private placement and bank term loans.)
-
The Alert Company is a closely held investment-services group that has been very successful over the past five years, consistently providing most members of the top management group with 50% bonuses....
-
Most of the Fischer esterification mechanism is identical with the mechanism of acetal formation. The difference is in the final step, where a carbocation loses a proton to give the ester. Write...
-
A carboxylic acid has two oxygen atoms, each with two nonbonding pairs of electrons. (a) Draw the resonance forms of a carboxylic acid that is protonated on the hydroxyl oxygen atom. (b) Compare the...
-
Show how Fischer esterification might be used to form the following esters. In each case, suggest a method for driving the reaction to completion. (a) Methyl salicylate (b) Methyl formate (bp 32 C)...
-
a) Give one reason why local Pakistan bus businesses could benefit from Pakistan's crisis. b) Give one reason why local bus businesses could be hurt by the Rupee crisis.
-
A P/E multiple could be high relative to the S&P 500 Index because its high growth rate justifies it. You notice a stock that has an assumed growth rate of 5.43% with a dividend payout ratio of 64.9%...
-
What would be the volume ( cm 3 ) of a piece of Styrofoam if it had a mass of 3 5 0 g , no matter its shape? Show calculation.

Study smarter with the SolutionInn App


