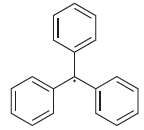
The triphenylmethyl radical was the first radical to be observed. Draw all resonance structures of this radical,
Question:
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (9 reviews)
This radica...View the full answer

Answered By

Ashok Kumar Malhotra
Chartered Accountant - Accounting and Management Accounting for 15 years.
QuickBooks Online - Certified ProAdvisor (Advance - QuickBooks Online for 3 years.
5.00+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Draw all resonance structures for each of the following radicals: (a) (b) (c) (d) (e)
-
Consider the structure of cyclopentadiene and then answer the following questions: (a) How many sp 3 -hybridized carbon atoms are present in the structure of cyclopentadiene? (b) Identify the most...
-
Draw the enolate ion that is formed when each of the following compounds is treated with sodium ethoxide. In each case, draw all resonance structures of the enolate ion, and predict whether a...
-
You have just been given a $454,000, which you decide to invest at an APR of 6.7 percent. If you were to withdraw $38,500 at the end of each year, starting at the end of this year, how many years...
-
Which of the following statements concerning the MM extension with growth is NOT CORRECT? a. The value of a growing tax shield is greater than the value of a constant tax shield. b. For a given D/S,...
-
Simpson should estimate the after-tax operating cash flow for Years 16 and 712, respectively, to be closest to: A. 0.31 billion and 0.21 billion. B. 0.31 billion and 0.25 billion. C. 0.35 billion and...
-
Describe the responsibilities of patients as reviewed in this chapter.
-
Casey Belzer runs a small machine shop that fabricates parts for sprayers used in foam insulation equipment. With the renewed interest in green building practices and high energy costs demand for his...
-
Custom Cabinetry has one job in process ( Job 1 2 0 ) as of June 3 0 ; at that time, its job cost sheet reports direct materials of $ 7 , 3 0 0 , direct labor of $ 3 , 8 0 0 , and applied overhead of...
-
V2x-x2 On converting into polar coordinates, (x +y?)dxdy is The mass of the lamina in the form of cardioids r = a(1 + cost) whose density varies as the square of the distance from the initial line is...
-
Calculate the pressure exerted by benzene for a molar volume of 2.00 L at 595 K using the RedlichKwong equation of state: The RedlichKwong parameters a and b for benzene are 452.0 bar dm 6 mol 2 K...
-
Use the equation C P,m C V ,m = TV m 2 / and the Data Tables to determine C V ,m for H 2 O(l) at 298 K. Calculate (C p,m C V,m )/C P,m .
-
In Exercises (a) Find the indefinite integral in two different ways. (b) Use a graphing utility to graph the antiderivative (without the constant of integration) obtained by each method to show that...
-
Goods are sold FOB destination. Tic Tac Corp. sold an air conditioner with a selling price of $10,200 to Cool Down Inc. The air conditioner was loaded on a delivery truck and shipped February 27, but...
-
I Spy With My LIttle Eyes produces designer sunglasses. Its projected financial information for the next period is as follows: Sales of 50,000 units @ $45 each for a total of $2,250,000 Total...
-
Chalice Ltd sells equipment to Chris Ltd on 1 Jul 2023. Chris Ltd qualifies for a 2-year interest free period. The current cash sales price is $40,000 and Chris Ltd will pay $40,000 on 30 June 2025....
-
Calculate what a landlord would receive in their rent disbursement based on the following scenario (please show your calculations). The landlords ledger account holds $1,500 rent and $120.55 for...
-
Angela and Frank are brother and sister and live in the same household. Angela has $50,000 of tax paid capital and she lends it to her brother to invest in his name because he is in a much lower tax...
-
Your friend Paul says that the helium used to inflate balloons is a product of radioactive decay. Your mutual friend Steve says no way. Then theres your friend Alison, who frets about living near a...
-
Which, if any, of the dichloroethene molecules drawn in Data Table II (3.) (4.) and (5.) are geometric isomers? A. B. C. D. cis-1,2-dichloroethene and trans-1,2-dichloroethene...
-
Propose structures for amines with the following 1H NMR spectra: (a) C3H9NO (b)C4H11NO2 TMS O ppm 10 6. Chemical shift (8) TMS O ppm 10 7. 9. Chemical shift (8) Intensity Intensity -3-
-
Propose structures for compounds that show the following 1H NMR spectra. (a) C9H13N (b)C15H17N TMS 10 6 5 3 2 O ppm Chemical shift (8) TMS 3 2 O ppm 10 6 1 Chemical shift (8) Intensity Intensity-
-
?-Amino acids can be prepared by the Strecker synthesis, a two-step process in which an aldehyde is treated with ammonium cyanide followed by hydrolysis of the amino nitrile intermediate with aqueous...
-
Discuss with examples about financial innovation& how it develops banking industry?
-
Elaborate with real world examples about different financial markets that contributed towards growth of financial sector?
-
The following statement of financial position is for the partnership of Able, Brown, and Crown at November 1, 2018. Assets Liabilities Cash $ 20,000 Accounts payable $ 50,000 Other assets 180,000...

Study smarter with the SolutionInn App


