Between 0°C and 100°C, the heat capacity of Hg(l) is given by Calculate ÎH and ÎS if
Question:
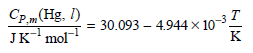
Calculate ΔH and ΔS if 2.25 moles of Hg(l) is raised in temperature from 0.00° to 88.0°C at constant P.
Transcribed Image Text:
Cp.„(Hg. I) 30.093 – 4.944 × 10-32 JK' mol K
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (12 reviews)
36115 AH n Cpmd TK 27315 225 mol 30093 ...View the full answer

Answered By

Susan Juma
I'm available and reachable 24/7. I have high experience in helping students with their assignments, proposals, and dissertations. Most importantly, I'm a professional accountant and I can handle all kinds of accounting and finance problems.
4.40+
15+ Reviews
45+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The standard entropy of Pb(s) at 298.15 K is 64.80 J K -1 mol - 1 . Assume that the heat capacity of Pb(s) is given by The melting point is 327.4C and the heat of fusion under these conditions is...
-
The heat capacity of α -quartz is given by The coefficient of thermal expansion is given by β = 0.3530 à 10 4 K 1 and V m = 22.6 cm 3 mol+. Calculate ÎS m for...
-
A 1.75 mole sample of an ideal gas is compressed isothermally from 62.0 L to 19.0 L using a constant external pressure of 2.80 atm. Calculate q, w, U, and H.
-
A 200 g mass attached to a horizontal spring oscillates at a frequency of 1.5 Hz. At one instant, the mass is at x = 70 mm and has vx = -0.2 m/s. Determine (a) The period (b) The amplitude (c) The...
-
Silva Piping Company produces PVC piping in two processing departments-Fabrication and Packaging. Transactions for the month of July are shown as follows. 1. Direct materials totaling $15,000 are...
-
Consider the same setting as problem 20, but suppose instead 80% of the shareholders redeem their shares, and no warrants are exercised. a. What is the amount of cash per share contributed by the...
-
Explain the relationship between culture and how people use symbols, rituals, and events to classify their world and create meaning.
-
On November 30, the end of the current fiscal year, the following information is available to assist Allerton Companys accountants in making adjusting entries: a. Allertons Supplies account shows a...
-
On January 1, 2021, TulipCo sells equipment it manufactured to PoppyCo in exchange for a $1 million noninterest-bearing note due in two years. The note bears no explicit interest. It says only that...
-
Some of the operations in standard programming languages can be modeled as actions that change the state of the world. For example, the assignment operation copies the contents of a memory location,...
-
The initiation step for radical addition of HBr is highly endothermic: (a) Explain how this step can be thermodynamically favorable at high temperature even though it is endothermic. (b) Explain why...
-
Draw all resonance structures for each of the following radicals: (a) (b) (c) (d) (e)
-
Find the area of the circle with the given radius or diameter. r = 45.8 cm
-
Define the term "prime cost." QB: A caf uses the prime cost pricing method and a price factor of 3.25. What is the preliminary menu price for a dish in this caf with a food cost per portion of $1.68...
-
most expensive quality cost category per unit of benefit obtained
-
Journals Look for six situations where you have prepared a dish for a customer who has a food allergy or intolerance. (Keep in mind that you should report on three food allergies and three...
-
The following information relates to Diamond Inc. Line Item Description Numerical Data Units sold ($500 per unit) 2,100 Variable costs per unit: Direct materials $90 Direct labor 60 Variable overhead...
-
prepare the adjusting journal entries elli dance company needs to make on their books to make the cash balance to agree with the adjusted book balance on the bank reconcilliation
-
Identify and sketch the graph of each relation. |(x 4)2, (y + 1) = 0 2. 00
-
What is your assessment of the negotiations process, given what you have studied? What are your recommendations for Mr. Reed? You must justify your conclusions
-
Repeat the calculation in Problem 11.4but plot the probability densities of the two orbitals. Then form the difference density, the difference between 2 and | 2a + 2b|
-
Imagine a small electron-sensitive probe of volume 1.00 pm3 inserted into an H+2 molecule-ion in its ground state. Calculate the probability that it will register the presence of an electron at the...
-
The same data as in Problem 11.8 may be used to calculate the molecular potential energy curve for the antibonding orbital, which is given by Plot the curve
-
How can the circular wait condition be detected and broken to prevent deadlock in complex systems with multiple interdependent resources ?
-
Case Study 1\ You have been appointed as a financial analyst for the Axis International Company, a profitable retail company. The director of Finance, belonging to the capital budgeting division has...
-
How can I find a rival hypothesis for my current thesis of Walmart's CEO should implement intra-team communication for all associates to help with communication barriers between managers and...

Study smarter with the SolutionInn App


