The chlorination of vinyl chloride, C 2 H 3 Cl + Cl 2 C 2 H 3
Question:
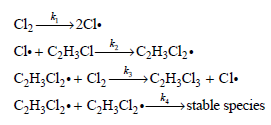
Derive the rate law expression for the chlorination of vinyl chloride based on this mechanism.
Transcribed Image Text:
Cl2-2C1. Cl. + C2H3CI- С Н.С1,-+ Cl, — сн,C + —Сн.Cl. Cl. →stable species СН.С1,.+ Снс1,.—
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 71% (7 reviews)
The rate of vinyl chloride production is The rate ...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The reaction I2(aq) + OCl2(aq) IO2(aq) + Cl2(aq) is believed to occur by the following mechanism: Write the rate law for this reaction.
-
The hydrogenbromine reaction corresponds to the production of HBr(g) from H 2 (g) and Br 2 (g) as follows: H 2 (g) + Br 2 (g) 2HBr(g). This reaction is famous for its complex rate law, determined by...
-
For the reaction I (aq) + OCl (aq) OI (aq) + Cl (aq) occurring in aqueous solution, the following mechanism has been proposed: a. Derive the rate law expression for this reaction based on this...
-
Calculate the following integral approximately by using the Simpson's method: 3.6 f. f(x) dx X 0.0 0.6 1.2 1.8 2.4 3.0 3.6 f(x) 0 0.13 0.05 0.6 0.43 0.74 0.93
-
The function f(x) = k(2 x x3) has an inverse function, and f1(3) = 2. Find k.
-
Intentional misstatement of a material fact. a. contract of adhesion b. duress c. fraud d. intentional c oncealment e. misrepresentation f. mistake g. mutual a greement h. puffi ng i. unconscionable...
-
Due to a nationwide recession, Amesbury Systems merchandise inventory is gathering dust. It is now October 31,2010, and the \($163,300\) that Amesbury Systems paid for its ending inventory is...
-
Companies are using the power of computing to develop continuous monitoring approaches to identify control problems early and to take corrective action on a timely basis. a. Explain how the concept...
-
Business Case Fat-E Bike You have been hired as the Director of Transportation for Fat-E. Fat-E is three-year-old firm, founded by three university friends based out of Ottawa, Ontario. The company...
-
Using Mohrs circle, determine for the cross section of the rolled-steel angle shown the orientation of the principal centroidal axes and the corresponding values of the moments of inertia....
-
Consider the collision-induced dissociation of N2O5(g) via the following mechanism: The asterisk in the first reaction indicates that the reactant is activated through collision. Experimentally it is...
-
In the unimolecular isomerization of cyclobutane to butylene, the following values for kuni as a function of pressure were measured: Assuming that the Lindemann mechanism accurately describes this...
-
A bank estimated that the standard error for a 95% confidence interval for the proportion of a certain demographic group that may default on a loan is 0.31. The lower confidence limit was calculated...
-
Watch The Deepest Hole in the World, and What We've Learned About It and complete the questions below by highlighting the correct answers: A. Choose the TRUE statement below. 1. Scientists were...
-
QUESTIONS NUMBERS ONE a) List five types of systems b) Explain the following characteristics of a system: (i) Differentiation; (ii) Equifinality; (iii) Feedback; (iv.) Transformation; (5 marks) (v.)...
-
You silver the inside of a half-sphere of radius 50cm to make aspherical mirror to use as a solar cooker. At what distance fromthe bottom of the ball should the food be placed for maximumheating?
-
For the two independent cases that follow, determine the missing amount for each letter. (Hint: You might not be able to calculate them in the order in which they appear.) Revenues Expenses Net...
-
When water falls upon snow it melts the snow due to the release of latent heat of fusion / solidification (80 cal/g). How much rain will it take (in centimeters) to melt 1.5 meter of snow in the...
-
In 2018, Bianca earned a salary of $164,000 from her employer, CallMart. How much in FICA and Medicare taxes will CallMart withhold from Biancas salary?
-
A handrail, which weighs 120 N and is 1.8 m long. was mounted to a wall adjacent to a small set of steps (Figure P4.26). The support at A has broken, and the rail has fallen about the loose bolt at 8...
-
Identify the missing reagent needed to achieve the following transformations: a. b. c. d. Br KF benzene Br NaF benzene
-
Consider the oxidation of the amino acid glycine NH 2 CH 2 COOH to produce water, carbon dioxide, and urea NH 2 CONH 2 : NH 2 CH 2 COOH(s) + 3O 2 (g) NH 2 CONH 2 (s) + 3CO 2 (g) + 3H 2 O(l)...
-
Show what reagents you would use to prepare each of the following ethers via a Williamson ether synthesis, and explain your reasoning. a. b. c. OMe
-
The following account balances are taken from the ledger of Maxwell Limited on 31 December 2018, the end of its fiscal year: Maxwell Limited Trial Balance As on 31 December 2018 S'm S'm Investment...
-
A researcher wishes to assess whether vitamin C is effective in the treatment of colds. To evaluate this hypothesis, the researcher decides to conduct a 2-year experimental study. The researcher...
-
Cullumber Co. acquired 25% of the 520,000 shares of outstanding common stock of Oriole Inc. on December 31, 2025. The purchase price was $3,913,000. Oriole declared and paid $1.20 per share cash...

Study smarter with the SolutionInn App


