In the unimolecular isomerization of cyclobutane to butylene, the following values for kuni as a function of
Question:
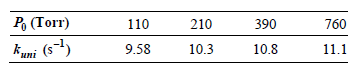
Assuming that the Lindemann mechanism accurately describes this reaction, determine k1 and the ratio k€“1/k2
Transcribed Image Text:
390 210 110 Po (Torr) 760 11.1 10.8 10.3 9.58 kuni (s-)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 42% (7 reviews)
The Lindemann mechanism predicts the following relationship between the unimol...View the full answer

Answered By

Allan Simiyu
I am an adroit Writer. I am a dedicated writer having worked as a writer for 3 years now. With this, I am sure to ace in the field by helping students break down abstract concepts into simpler ideas.
5.00+
8+ Reviews
54+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Consider the unimolecular isomerization of methylcyanide, a reaction that will be discussed in detail in Chapter 36: CH 3 NC(g) CH 3 CN(g) The Arrhenius parameters for this reaction are A = 2.5 10...
-
In Problem 22.10 the isomerization of cyclopropane over a limited pressure range was examined. If the Lindemann mechanism of first order reactions is to be tested we also need data at low pressures....
-
The isomerization of cyclopropane, C3H6, to propylene, CH2=CHCH3, is first order in cyclopropane and first order overall. At 1000oC, the rate constant is 9.2/s. What is the half-life of cyclopropane...
-
k) Assume that one of these portfolio's is the Market Portfolio and all portfolios, except Portfolio G, are fairly priced according to the CAPM. What is the highest utility score that can be achieved...
-
Prove that if f is a one-to-one odd function, then f1 is an odd function.
-
Which aspects of your business should you keep inhouse and which should you outsource? How do you protect your competitive advantage?
-
The equilibrium constant of a chemical reaction is influenced by the (a) Pressure (b) Temperature (c) Initial concentration of the reacting substance (d) None of these.
-
Bexar Companys fixed overhead costs for the year are expected to be as follows: depreciation, $80,000; supervisory salaries, $90,000; property taxes and insurance, $25,000; and other fixed overhead,...
-
You work for the law firm Skywalker, Solo, & Organa LPA. They're a small firm that represents Channel 5, the local NBC affiliate. Channel 5 is hiring a new prime-time news anchor and they've asked...
-
In HTTP, draw a figure to show the application of cookies in a web portal using two sites.
-
The chlorination of vinyl chloride, C 2 H 3 Cl + Cl 2 C 2 H 3 Cl 3 , is believed to proceed by the following mechanism: Derive the rate law expression for the chlorination of vinyl chloride based on...
-
In the discussion of the Lindemann mechanism, it was assumed that the rate of activation by collision with another reactant molecule, A, was the same as collision with a nonreactant molecule, M, such...
-
In problem, draw each angle. 21/4
-
Describe the methodologies used for whole-genome sequencing and the challenges associated with interpreting complex genotypic data to predict potential phenotypic outcomes .
-
Exotic Engine Shop uses a job order cost system to determine the cost of performing engine repair work. Estimated costs and expenses for the coming period are as follows: Engine parts $1,356,300 Shop...
-
How do variations in the genotype lead to different phenotypic expressions, and what are the roles of epigenetic modifications in modulating these relationships ?
-
Total overhead of $235,000 is allocated according to the proportion of labour hours. Department A used 1830 hours, department B used 1635 hours, and department C used 1760 hours. What amount of...
-
In this activity, you will download two tables of data from the Homeland Infrastructure Foundation-Level Data (HIFLD) website and one table of data from the American Community Survey (ACS)....
-
A contractor constructs homes with 5 different choices of exterior finish, 3 different roof arrangements, and 4 different window designs. How many different types of homes can be built?
-
Activator rod AB exerts on crank BCD a force P directed along line AB. Knowing that P must have a 100-N component perpendicular to arm BC of the crank, determine (a) The magnitude of the force P, (b)...
-
Calculate the average CH bond enthalpy in methane using the data tables. Calculate the percent error in equating the average CH bond energy in Table 4.3 with the bond enthalpy. Table 4.3 Selected...
-
The heat capacity of α -quartz is given by The coefficient of thermal expansion is given by β = 0.3530 à 10 4 K 1 and V m = 22.6 cm 3 mol+. Calculate ÎS m for...
-
The following cyclic ether can be prepared via an intramolecular Williamson ether synthesis. Show what reagents you would use to make this ether.
-
Ridge Crest Company has beginning Retained Earnings of $23,000, ending Retained Earnings of $32,600, and net income of $15,500. What was the amount of dividends declared during the year?
-
Solomon Manufacturing Company established the following standard price and cost data. Sales price Variable manufacturing cost Fixed manufacturing cost $ 8.50 per unit Fixed selling and administrative...
-
Shadee Corporation expects to sell 640 sun shades in May and 380 in June. Each shade sells for $134. Shadee's beginning and ending finished goods inventories for May are 70 and 40 shades,...

Study smarter with the SolutionInn App


