Use the vapor pressures of ice given here to calculate the enthalpy of sublimation using a graphical
Question:
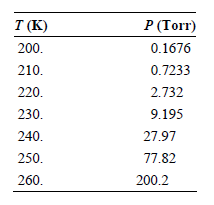
Transcribed Image Text:
T (K) P (Torr) 200. 0.1676 210. 0.7233 2.732 220. 230. 9.195 240. 27.97 250. 77.82 200.2 260.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (6 reviews)
A least squares fit of ...View the full answer

Answered By

JAPHETH KOGEI
Hi there. I'm here to assist you to score the highest marks on your assignments and homework. My areas of specialisation are:
Auditing, Financial Accounting, Macroeconomics, Monetary-economics, Business-administration, Advanced-accounting, Corporate Finance, Professional-accounting-ethics, Corporate governance, Financial-risk-analysis, Financial-budgeting, Corporate-social-responsibility, Statistics, Business management, logic, Critical thinking,
So, I look forward to helping you solve your academic problem.
I enjoy teaching and tutoring university and high school students. During my free time, I also read books on motivation, leadership, comedy, emotional intelligence, critical thinking, nature, human nature, innovation, persuasion, performance, negotiations, goals, power, time management, wealth, debates, sales, and finance. Additionally, I am a panellist on an FM radio program on Sunday mornings where we discuss current affairs.
I travel three times a year either to the USA, Europe and around Africa.
As a university student in the USA, I enjoyed interacting with people from different cultures and ethnic groups. Together with friends, we travelled widely in the USA and in Europe (UK, France, Denmark, Germany, Turkey, etc).
So, I look forward to tutoring you. I believe that it will be exciting to meet them.
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Use the vapor pressures of SO 2 (l) given in the following table to calculate the enthalpy of vaporization using a graphical method or a least squares fitting routine. T (K) 190. P (Pa) T (K) 230. P...
-
Use the vapor pressures of n-butane given in the following table to calculate the enthalpy of vaporization using a graphical method or a least squares fitting routine. P (Pa) 1000 x 104 1000 x 105 T...
-
Use the vapor pressures of tetrachloromethane given in the following table to calculate the enthalpy of vaporization using a graphical method or a least squares fitting routine. P/Pa T (K) 320. 330....
-
A Norman window has the outline of a semicircle on top of a rectangle. Suppose there are 8 + feet of wood trim available. Discuss why a window designer might want to maximize the area of the window....
-
Select 4 similar size public companies in the same industry. The company's size can be measured by total assets or total sales. Obtain the most recent financial statements of these companies, apply...
-
How can one adjust the venture capital valuation method to account for debt?
-
The Eaton Company was incorporated July 10, 1999, with authorized capital as follows: a. Common stock. Class A, 20,000 shares, par value $25 per share. b. Common stock. Class B, 100,000 shares, par...
-
Boxer Incorporated has hired you as a consultant to implement a data-driven fraud detection program in the company. One of the owners, Priscilla Boxer, asks you to give a presentation to several...
-
how does Apple use the agile organizational model to adapt and change to changing situations without traditional change management interventions. Explain.
-
How should Nick Swinmurn, Zappos founder, be described in terms of the qualities of a founder that are discussed in this chapter?
-
The phase diagram of NH 3 can be characterized by the following information. The normal melting and boiling temperatures are 195.2 and 239.82 K, respectively; the triple point pressure and...
-
Calculate the vapor pressure for a mist of spherical water droplets of radius a. 1.95 10 8 m b. 2.25 10 6 m at 298 K. The vapor pressure of water at this temperature is 25.2 Torr.
-
Find the derivatives of the functions in Problem. y = 4 x 2
-
You buy a nine-year bond for $940. The bond has a 3.10% coupon rate. Coupons are s/a. What is the bond's yield to maturity?
-
A company is expected to grow earnings by 4.2% per year forever. The firm's cost of equity is 9.2% and the firm's ROE is 12%. What should the firm's forward P/E be?
-
The Melbourne Corp. wants to set up a private holiday business. According to the CFO, Larry Sheep. business is booming. As a result, the project will provide a net cash inflow of $200,000 for the...
-
Earnhardt Driving School's 2017 balance sheet showed net fixed assets of $3.1 million, net working capital of 1 million, and long-term debt of 0.5 million. The 2018 balance sheet showed net fixed...
-
a) b) Compare and contrast American options on forwards and futures with European options, and identify the appropriate pricing model for European options. (9 Marks) Evaluate the three primary ways...
-
Use (a) The Trapezoidal Rule, (b) The Midpoint Rule, and (c) Simpsons Rule to approximate the given integral with the specified value of n. (Round your answers to six decimal places.) e* dx, %3D 10...
-
Consider the reaction of acetic acid in water CH 3 CO 2 H(aq) + H 2 O(l) CH3CO 22 (aq) + H 3 O + (aq) where Ka 5 1.8 3 1025. a. Which two bases are competing for the proton? b. Which is the stronger...
-
Find the gradient of the function g(x,y,z) = ax 3 + ye bz , where a and b are constants.
-
Find r where r = ix + jy + kz.
-
Find the Laplacian of the function f = exp (x 2 + y 2 + z 2 ) = e x2 e y2 e z2 .
-
Analyze the following problems in JMP and answer the questions below: GPA Hours TV 3.24 19 3.1 21 3.24 18 2.61 30 3.03 23 3.37 10 1.6 36 3.2 10 2.96 13 2.18 31 2.5 33 2.95 4 2.72 25 3.05 21 3.45 8...
-
Leadership in a Multicultural Tech Company Company: Tech Vista Innovations Inc. Industry: Information Technology Background: Tech Vista Innovations is a fast-growing global tech company that...
-
Define the following terms: capital goods, consumer goods and services, consumption, demand, distribution, economic goods, economic resources, economic services, economic need, economy, elastic...

Study smarter with the SolutionInn App


