Classify bond as ionic or covalent from formula. (Pool 1 of 2) Determine whether bonds in...
Fantastic news! We've Found the answer you've been seeking!
Question:



Transcribed Image Text:
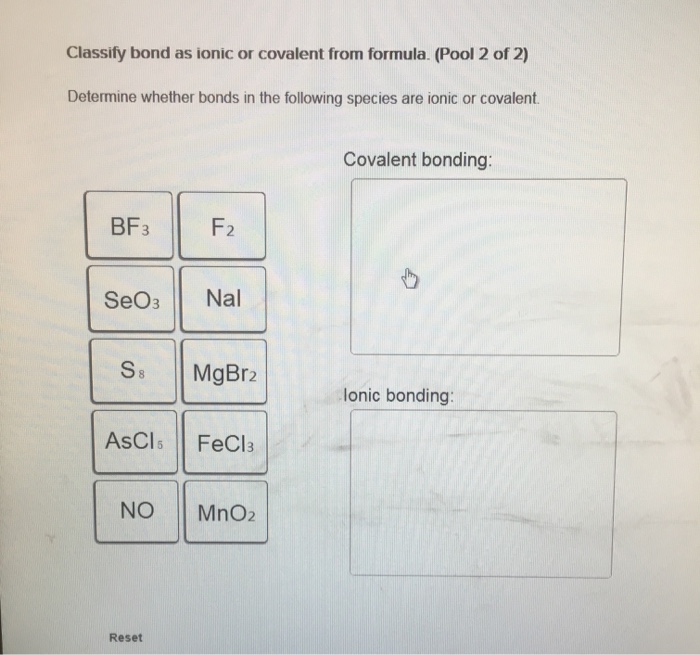
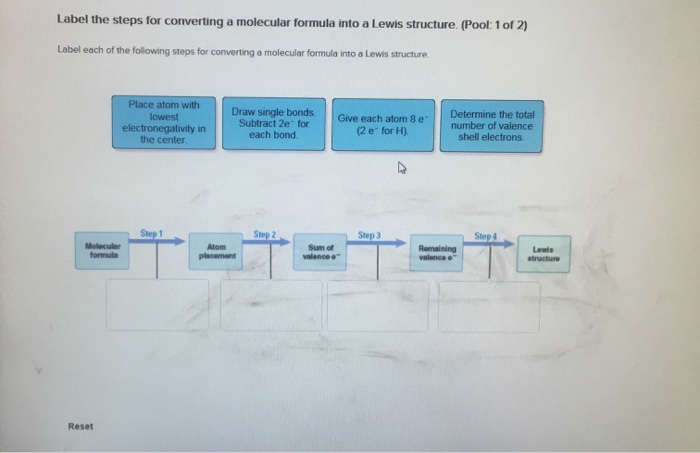
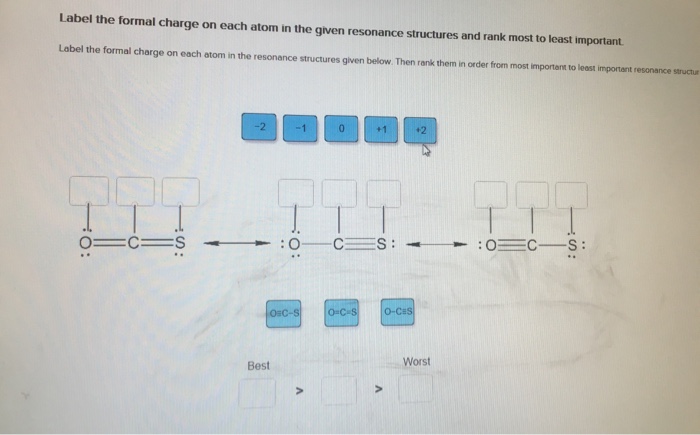
Classify bond as ionic or covalent from formula. (Pool 1 of 2) Determine whether bonds in the following species are ionic or covalent Cl₂ CO CaF2 CrO2 HF Reset References Classification CaO P4 KCI SiCl4 CUS Covalent bonding: lonic bonding: Classify bond as ionic or covalent from formula. (Pool 2 of 2) Determine whether bonds in the following species are ionic or covalent. BF3 SeO3 S8 ASCI 5 NO Reset F2 Nal MgBr2 FeCl3 MnO2 Covalent bonding: D lonic bonding: Complete and sequence the steps for drawing Lewis structures. Complete and correctly sequence the steps for drawing Lewis structures. octet add multiple core lone surrounding four single six singles lower Reset pairs one bonding subtract central valence Zoom less eight more two higher wwwwwwwww AAAAAAAAAAAwww Drag the text blocks below into their correct order. Distribute the remaining electrons as up with electrons (or pairs on atoms to give each an the electrons If the make a Determine the total number of For polyatomic ions, charge, or from one of the the Draw a central atom, and bond. atom. Then check that each atom has so that each atom ends for H). First place electronegative) atom. If any electrons remain, place them on electrons available. one electron for each negative one electron for each positive charge. atom still does not have a(n) bond by changing a atoms into a bond from each surrounding atom to the two valence electrons for each Place the atoms relative to each other. Place the atom with number, place the atom with the the center. pair pair to group number in the center because it has the electronegativity. If atoms have the same group period number in Label the steps for converting a molecular formula into a Lewis structure. (Pool: 1 of 2) Label each of the following steps for converting a molecular formula into a Lewis structure. Place atom with lowest electronegativity in the center. Molecular formula Reset Step 1 Draw single bonds. Subtract 2e for each bond. Atom placement Step 2 Sum of valence o Give each atom 8 e (2 e for H). Step 3 Determine the total number of valence shell electrons. Remaining valence Step 4 Lowie structure 14. value: 6.52 points Step 1 Label the steps for converting a molecular formula into a Lewis structure. (Pool: 2 of 2) Label each of the following steps for converting a molecular formula into a Lewis structure. Remaining valence e < Place atom with lowest EN in center Question 14 (of 23) Sum of valence e Molecular formula Step 2 Total the number of valence shell electrons Lewis Structure Step 3 Draw single bonds, and subtract 20 for each bond Atom placement Step 4 Give each atom Be (2e" for H) Label the formal charge on each atom in the given resonance structures and rank most to least important. Label the formal charge on each atom in the resonance structures given below. Then rank them in order from most important to least important resonance structur 0=C=S -2 -1 Best OSC-S 0 > :0 CES: - +1 O-C-S A +2 O-CES Worst :0 CS: Classify bond as ionic or covalent from formula. (Pool 1 of 2) Determine whether bonds in the following species are ionic or covalent Cl₂ CO CaF2 CrO2 HF Reset References Classification CaO P4 KCI SiCl4 CUS Covalent bonding: lonic bonding: Classify bond as ionic or covalent from formula. (Pool 2 of 2) Determine whether bonds in the following species are ionic or covalent. BF3 SeO3 S8 ASCI 5 NO Reset F2 Nal MgBr2 FeCl3 MnO2 Covalent bonding: D lonic bonding: Complete and sequence the steps for drawing Lewis structures. Complete and correctly sequence the steps for drawing Lewis structures. octet add multiple core lone surrounding four single six singles lower Reset pairs one bonding subtract central valence Zoom less eight more two higher wwwwwwwww AAAAAAAAAAAwww Drag the text blocks below into their correct order. Distribute the remaining electrons as up with electrons (or pairs on atoms to give each an the electrons If the make a Determine the total number of For polyatomic ions, charge, or from one of the the Draw a central atom, and bond. atom. Then check that each atom has so that each atom ends for H). First place electronegative) atom. If any electrons remain, place them on electrons available. one electron for each negative one electron for each positive charge. atom still does not have a(n) bond by changing a atoms into a bond from each surrounding atom to the two valence electrons for each Place the atoms relative to each other. Place the atom with number, place the atom with the the center. pair pair to group number in the center because it has the electronegativity. If atoms have the same group period number in Label the steps for converting a molecular formula into a Lewis structure. (Pool: 1 of 2) Label each of the following steps for converting a molecular formula into a Lewis structure. Place atom with lowest electronegativity in the center. Molecular formula Reset Step 1 Draw single bonds. Subtract 2e for each bond. Atom placement Step 2 Sum of valence o Give each atom 8 e (2 e for H). Step 3 Determine the total number of valence shell electrons. Remaining valence Step 4 Lowie structure 14. value: 6.52 points Step 1 Label the steps for converting a molecular formula into a Lewis structure. (Pool: 2 of 2) Label each of the following steps for converting a molecular formula into a Lewis structure. Remaining valence e < Place atom with lowest EN in center Question 14 (of 23) Sum of valence e Molecular formula Step 2 Total the number of valence shell electrons Lewis Structure Step 3 Draw single bonds, and subtract 20 for each bond Atom placement Step 4 Give each atom Be (2e" for H) Label the formal charge on each atom in the given resonance structures and rank most to least important. Label the formal charge on each atom in the resonance structures given below. Then rank them in order from most important to least important resonance structur 0=C=S -2 -1 Best OSC-S 0 > :0 CES: - +1 O-C-S A +2 O-CES Worst :0 CS:
Expert Answer:

Related Book For 

Chemistry The Central Science
ISBN: 978-0321696724
12th edition
Authors: Theodore Brown, Eugene LeMay, Bruce Bursten, Catherine Murphy, Patrick Woodward
Posted Date:
Students also viewed these chemistry questions
-
Classify each of the labeled bonds in the following structure in terms of the bond type ( or ) and the component orbitals that overlap to form the bond. (For example, the carbon--carbon bond in...
-
Predict the bond angles for all bonds in the following compounds: a) CH 3 CH 2 OH b) CH 2 O c) C 2 H 4 d) C 2 H 2 e) CH 3 OCH 3 f) CH 3 NH 2 g) C 3 H 8 h) CH 3 CN
-
Which amide bonds in the following polypeptide are cleaved by trypsin by chymotrypsin? Phe-Leu-Met-Lys-Tyr-Asp-Gly-Gly-Arg-Val-IIe- Pro-Tyr
-
Pricing in imperfect markets (continuation of 22-28). Refer to Problem 22-28. 1.Suppose the manager of Division A has the option of (a) cutting the external price to $195, with the certainty that...
-
What is the status of the Export Administration Act of 1979 and the Export Administration Regulations? Has the statute been renewed or replaced since its lapse in 2001? Are the regulations still in...
-
Estimate the vapor pressure of acetone (mm Hg) at 50C (a) from data in Perrys Chemical Engineers Handbook (Footnote 1) and the ClausiusClapeyron equation, (b) from the Antoine equation using...
-
Is there a relationship between the number of accounting standards issued and demand for accounting and auditing services? Explain this relationship. Given this relationship, should environmental...
-
Can Pepsi or any other company ever surpass Coca- Cola? Why or why not? What are Coca-Colas greatest risks?
-
Handy Hardware is a retail hardware store. Information about the store's operations follows. November 20x1 sales amounted to $200,000. Sales are budgeted at $220,000 for December 20x1 and $210,000...
-
The cash account for Highlander Diamond Co. on April 1, 20Y5, indicated a balance of $35,400. During April, the total cash deposited was $120,255, and checks written totaled $109,200. The bank...
-
A) Company C is financed only through equity and straight debt. The company has conflicts of interest between shareholders, on one hand, and the outstanding debtholders, on the other hand. Would an...
-
Consider an object of characteristic length \(0.015 \mathrm{~m}\) and a situation for which the temperature difference is \(10^{\circ} \mathrm{C}\). Evaluating thermophysical properties at the...
-
What is the convection mass transfer coefficient associated with fully developed atmospheric airflow at \(27^{\circ} \mathrm{C}\) and \(0.04 \mathrm{~kg} / \mathrm{s}\) through a...
-
As discussed in Section 5.2, the lumped capacitance approximation may be applied if \(B_{i} <0.1\), and, when implemented in a conservative fashion for a long cylinder, the characteristic length is...
-
An ideal gas flows within a small diameter tube. Derive an expression for the transition density of the gas \(ho_{c}\) below which microscale effects must be accounted for. Express your result in...
-
Consider a concentric tube annulus for which the inner and outer diameters are 25 and \(50 \mathrm{~mm}\). Water enters the annular region at \(0.03 \mathrm{~kg} / \mathrm{s}\) and \(25^{\circ}...
-
You are conducting a research project that seeks to understand the effect of age and attitude towards the brand on intention to purchase Red Bull products. Specify a research question and two...
-
A horizontal annulus with inside and outside diameters of 8 and 10 cm, respectively, contains liquid water. The inside and outside surfaces are maintained at 40 and 20oC, respectively. Calculate the...
-
A pound of plain M&M candies contains 96 g fat, 320 g carbohydrate, and 21 g protein. What is the fuel value in kJ in a 42-g (about 1.5 oz) serving? How many Calories does it provide?
-
Consider the combustion of a single molecule of CH4(g) forming H2O(l) as a product. (a) How much energy, in J, is produced during this reaction? (b) A typical X-ray light source has an energy of 8...
-
The coordination number for Mg2+ ion is usually six. Assuming this assumption holds, determine the anion coordination number in the following compounds: (a) MgS, (b) MgF2, (c) MgO.
-
Refer to the financial statements of Best Buy in Appendix A near the end of the book. Look at the cad consolidated statements of earnings (income statement).How many years are included and what are...
-
Accounting is an information and measurement system that ____________information about an organizations business activities. a. Translates b. Records c. Chooses d. Prints out
-
External users of financial information include: a. Purchasing managers b. Service managers c. The chief executive officer d. Lenders

Study smarter with the SolutionInn App


