Determine the crystal system and point-group symmetry from the space-group symbol for each of the following borates.
Question:
Determine the crystal system and point-group symmetry from the space-group symbol for each of the following borates. In each case determine the point group and state whether the symmetry permits SHG activity.
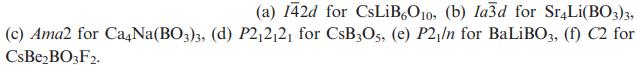
Transcribed Image Text:
(a) 142d for CsLiB6O10, (b) la3d for Sr4Li(BO3)3, (c) Ama2 for Ca4Na(BO3)3, (d) P21212 for CsB3O5, (e) P2/n for BaLiBO3, (f) C2 for CsBeBO3F2.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)
a Tetragonal point group 42m SHG allowed b Cubic po...View the full answer

Answered By

Muhammad Haroon
More than 3 years experience in teaching undergraduate and graduate level courses which includes Object Oriented Programming, Data Structures, Algorithms, Database Systems, Theory of Automata, Theory of Computation, Database Administration, Web Technologies etc.
5.00+
3+ Reviews
10+ Question Solved
Related Book For 

Solid State Materials Chemistry
ISBN: 9780521873253
1st Edition
Authors: Patrick M. Woodward, Pavel Karen, John S. O. Evans, Thomas Vogt
Question Posted:
Students also viewed these Sciences questions
-
For each of tire following precedence networks, determine the critical path and the project duration by determining the length of each path. The numbers above the nodes represent activity durations...
-
Managing Scope Changes Case Study Scope changes on a project can occur regardless of how well the project is planned or executed. Scope changes can be the result of something that was omitted during...
-
The prototypes and diagrams that we have created have really given us a feel for the project, smiles Anna, looking up from her computer. I have a good sense about what we are doing. Same here,...
-
Being employed as a casual barista in a local caf with a flourishing catering business called 'Brown's Caf' , where you are supervised by the caf owner, Jessica Williams. The company employs 20...
-
Bar AB rests on the two quarter-circle surfaces shown. A force P is applied at point C which is located at a distance a from end A. Neglecting the weight of the bar and knowing that the coefficient...
-
Suppose a time-reversible Markov chain has transition matrix P and stationary distribution . Show that the Markov chain can be regarded as a random walk on a weighted graph with edge weights w(i, j)...
-
In 1951, DuPont began using the chemical perfluorooctanoic acid to manufacture Teflon. Due to the dangerous nature of the chemical, DuPont was given special instructions by its supplier to dispose of...
-
The Palmer Acres Inn is trying to determine its break-even point during its off-peak season. The inn has 50 rooms that it rents at $60 a night. Operating costs are as follows. Salaries...
-
It is March, and the annual NCAA Basketball Tournament is down to the final 4 teams. Randy Kitchell is a t-shirt vendor who plans to order t-shirts with the names of the final 4 teams from a...
-
For borates with similar orientation of the BO 3 groups, the NLO coefficients should roughly scale with the coplanar character and density of the BO 3 groups. The following borates have highly...
-
In 2005, two new polymorphs of BiB 3 O 6 (- and -BiB 3 O 6 ) were synthesized using boric acid as a flux [25]. All three polymorphs adopt closely related monoclinic structures. While a-BiB306 has C2...
-
Pam Erickson Construction Company changed from the completed-contract to the percentage-of-completion method of accounting for long-term construction contracts during 2021. For tax purposes, the...
-
The hull of a boat has a volume of 180 m, and the total mass of the boat when empty is 8610 kg. Take the specific gravity of the sea water to be 1.03 and take the density of water to be 1000 kg/m....
-
You establish a straddle on Walmart using September call and put options with a strike price of $68. The call premium is $5.15 and the put premium is $5.90. a. What is the most you can lose on this...
-
Prestige Company has determined the following information for its recent fiscal year. Days inventory outstanding 34.2 days Days payable outstanding 45.4 days Days sales outstanding 73 days Compute...
-
Durable Metals Corporation holds an 80% equity stake in Green Mining Incorporated. On January 1, 20X1, Durable Metals sold land costing $300,000 to Green Mining for $360,000. On December 31, 20X1,...
-
The year-end financial statements for North Railway report the following information: Year ended December 31, (In millions) Year 2 Year 1 Revenues $19,829 $21,967 Property and equipment, net 49,000...
-
Tim Mattke Company began operations in 2006 and for simplicity reasons, adopted weighted-average pricing for inventory. In 2008, in accordance with other companies in its industry, Mattke changed its...
-
Can partitioned join be used for r r.A s? Explain your answer
-
There are two stereoisomers of 1-tert-butyl-4-chlorocyclohexane. One of these isomers reacts with sodium ethoxide in an E2 reaction that is 500 times faster than the reaction of the other isomer....
-
Arrange the following alkenes in order of increasing stability:
-
For each pair of the following compounds identify which compound would react more rapidly in an E1 reaction. a. b. CI .CI CI CI
-
For the below Repo transaction, calculate the Margin Ratio: Market value of collateral = BHD 2 million Amount of cash loan BHD 1.5 million = Accrued Interest = 0.2 million
-
It should not be a surprise to learn that nearly everyone has experienced cognitive dissonance after making a purchase of some kind. Describe the event and explain what you did about it. Discuss what...
-
Your company has been contracted to manage the construction of a new apartment complex. The project has a tight budget and timeline, and the client is concerned about the project being completed on...

Study smarter with the SolutionInn App


