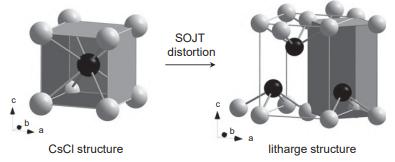
PbO adopts the tetragonal litharge structure that can be described as a distorted variant of the CsCl
Question:
PbO adopts the tetragonal litharge structure that can be described as a distorted variant of the CsCl structure:

Equation 5.18
![]()
Transcribed Image Text:
b a CsCl structure SOJT distortion litharge structure
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)
a The bondvalence equation gives dPbO 263 Twice this distance is the body diago...View the full answer

Answered By

Vijesh J
My passion to become a tutor is a lifetime milestone. Being a finance and marketing professional with hands-on experience in wealth management, portfolio management, team handling and actively contributing in promoting the company. Highly talented in managing and educating students in most attractive ways were students get involved. I will always give perfection to my works. Time is the most important for the works and I provide every answer on time without a delay. I will proofread each and every work and will deliver a with more perfection.
4.70+
5+ Reviews
15+ Question Solved
Related Book For 

Solid State Materials Chemistry
ISBN: 9780521873253
1st Edition
Authors: Patrick M. Woodward, Pavel Karen, John S. O. Evans, Thomas Vogt
Question Posted:
Students also viewed these Sciences questions
-
The structure of manganese fluoride can be described as a simple cubic array of manganese ions with fluoride ions at the center of each edge of the cubic unit cell. What is the charge of the...
-
A baseball game consists of plays that can be described as follows: Play Description No advance An out where no runners advance. This includes strikeouts, pop ups, short flies, and the like....
-
A baseball game consists of plays that can be described as follows: Distributions for these plays for two teams, the White Sox (visitors) and the Yankees (home), are asfollows: Team: White Sox Team:...
-
Pick a Nigerian brand that has been very successful competing against the bigger global brands in its product category. What are the elements of its strategy that have enabled it to achieve this...
-
As shown in Fig. P2.44, the 6-in-thick exterior wall of a building has an average thermal conductivity of 0.32Btu/h ft oR. At steady state, the temperature of the wall decreases linearly from T1 =...
-
Financial information for Robinson Inc. follows: Additional information: Ordinary shares were redeemed during the year at their book value. The face value of the bonds is $400,000; they pay a coupon...
-
Acne Medication and Gastrointestinal Injury Since 1999, Kamie Kendall had experienced ongoing abdominal pain, and she was eventually diagnosed with ulcerative colitis and irritable bowel syndrome...
-
During 2014, the Town of Falmouth had a number of transactions that affected net position of its town skating rink, which is operated as an enterprise fund. You are provided with the following...
-
5.) A hoop of 6 kg and radius .15 m is rolling down a 40-degree incline with 5 m length, what acceleration does it travel down the incline? How long does it take to get down the incline?
-
The black modification of SnO is isostructural with PbO litharge of the previous problem. (a) The bond-valence parameters for Sn 2+ O are R 0 = 1.98 and B = 0.37 . Use Equation (5.18) (d ij = R 0 ij...
-
BiAlO 3 and BiGaO 3 can be prepared by high-pressure synthesis. The space group of BiAlO 3 is R3c while that of BiGaO 3 is Pcca. Based on the symmetry alone, what can you say about the possibility...
-
Discuss how GIS-based, site-specific nutrient-application technology might improve profitability and reduce environmental degradation.
-
As discussed in the article "Federal Corporate Law: Lessons from History" one political impediment to sweeping federal change is the presumption in favor or state law in corporate matters. How might...
-
The Summit at Crystal Mountain is a small ski resort located just outside Mount Rainier, Washington; approximately 100 miles from Seattle. The resort consists of slopes that appeal to skiers of...
-
John has a job selling souvenirs at a football stadium. He earns $10 per game plus $0.25 for each souvenir he sells. How many souvenirs does John need to sell to earn a total of $35 for working at...
-
To support herself while attending school, Daun Deloch sold stereo systems to other students. During the first year of operations, Deloch purchased the stereo systems for $163,000 and sold them for...
-
It is important for all to understand what sources of data and statistical tools are available. Your assignment is to research and identify what tools are used frequently within your organization....
-
The accountant of Kooks Shoe Co. has compiled the following information from the companys records as a basis for an income statement for the year ended December 31, 2008. Rental revenue...
-
What is the amount of total interest dollars earned on a $5,000 deposit earning 6% for 20 years?
-
For each of the following descriptions draw the structure of a compound that fits the description. (Note: There are many correct answers for each of these problems.) a) An alkyl halide that produces...
-
Assuming that H f is constant in the interval 275 K 600. K, calculate G for the process *H2O, g, 298 K) (H 2 O, g, 600, K). Calculate the relative change in the Gibbs energy.
-
How many different alkenes will be produced when each of the following substrates is treated with a strong base? a) 1-Chloropentane b) 2-Chloropentane c) 3-Chloropentane d) 2-Chloro-2-methylpentane...
-
Compare the costs per unit of Job Order 410 and 411. Explain why the cost per unit under the traditional costing system is different from the cost per unit under ABC method.
-
Do the following contracts need to be in writing to be enforceable? Explain why or why not. A contract whereby the president of NewCo. Gives a personal guarantee (i.e., uses personal assets as...
-
What is merchantability? What is an example of merchantability?

Study smarter with the SolutionInn App


