Show that (10.8.5) is correct by performing the average of the exchange energy (8.12.3) over all k.
Question:
Show that (10.8.5) is correct by performing the average of the exchange energy (8.12.3) over all k.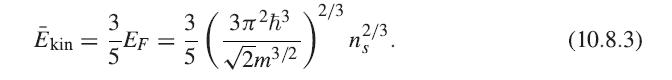
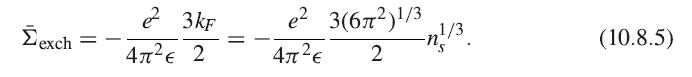
Transcribed Image Text:
2/3 3 3 Exin = ²-Er = ² ( 323/12) EF 5 5 n²/3. (10.8.3)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (4 reviews)
In order to prove the statement we have to calculate the ...View the full answer

Answered By

Rodrigo Louie Rey
I started tutoring in college and have been doing it for about eight years now. I enjoy it because I love to help others learn and expand their understanding of the world. I thoroughly enjoy the "ah-ha" moments that my students have. Interests I enjoy hiking, kayaking, and spending time with my family and friends. Ideal Study Location I prefer to tutor in a quiet place so that my students can focus on what they are learning.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The following additional information is available for the Dr. Ivan and Irene Incisor family from Chapters 1-5. Ivan's grandfather died and left a portfolio of municipal bonds. In 2012, they pay Ivan...
-
Planning is one of the most important management functions in any business. A front office managers first step in planning should involve determine the departments goals. Planning also includes...
-
The following additional information is available for the Dr. Ivan and Irene Incisor family from Chapters 1-4. Ivan and Irene paid the following in 2012 (all by check or can otherwise be...
-
41.(6 pointa) In the following balanced reaction, magnesium metal (Mg) reacts with carbon dioxide gas (CO) o form solid magnesium exide (MgO) and solid carbon (C). How many grams of carbon dioxide...
-
Almaden Hardware Store sells two distinct types of products, tools and paint products. Information pertaining to its 2011 year-end inventory is as follows: Required: 1. Determine the balance sheet...
-
Use a computer to develop the equation of the regression model for the following data. Comment on the regression coefficients. Determine the predicted value of y for x1 = 200 and x2 =7. 3948925185...
-
An 800-kg car moving at \(108 \mathrm{~km} / \mathrm{hr}\) hits and gets entangled with a large truck of inertia \(10,000 \mathrm{~kg}\) initially at rest. What is the kinetic energy of the wreckage?...
-
The ledger of Rowen Company contains the following balances: Retained Earnings $30,000; Dividends $2,000; Service Revenue $47,000; Salaries and Wages Expense $27,000; and Supplies Expense $5,000....
-
Write one page about pipelining in smartphones that support Android operating systems. (10 marks)
-
For T < T c , we can write m(x(vector)) = m 0 + m, where m 0 is the mean-field solution of the Ising model. Show that for h = 0, Gaussian fluctuations around the ordered state m 0 lead to a divergent...
-
Compute the specific heat C v = U/T, with U = Z/ = (F)/, for the Ising model in the cases T < T c and T > T c and show that there is a discontinuity at T c .
-
The graph shows a model for the number of millions of Social Security beneficiaries projected to 2030. The model was developed with data from the Social Security Trustees Report. (a) Was the...
-
What is the Marketing budget? Who is the hypothetical target audience? What are the key elements for the visual aspect of the campaign?
-
Kendrick discusses outsourcing and the procurement process. Identify and describe the four components of the procurement process as specified in PMI's PMBOK. Can any of these four components carry a...
-
Thunder Hollow Inc. makes and sells hood ornaments for $118 each. The company is considering upgrading its machinery, which would increase its fixed costs to a total of $28,000. However, the new...
-
The graph below depicts a government intervention setting at a price ceiling of $1,500 per month for a rental apartment. What is the value for the deadweight loss in this market? Price (monthly rent)...
-
For the proposed project (B above), identify and describe aspects of the project that would relate to the four values of the PMI Code of Ethics and Professional Conduct. How should the project...
-
Is ocranoic acid more soluble in 1 M HC1, 1 M NaOH, or pure water? Explain. Drugs such as morphine are often treated with strong acids. The most commonly used form of morphine is morphine...
-
What does non-recourse financing mean?
-
We saw a general rule that the two protons of a CH 2 group will be chemically equivalent if there are no chirality centers in the compound. An example of an exception is 3-bromopentane. This compound...
-
Identify the structure of a compound with molecular formula C 9 H 20 that exhibits four CH 2 groups, all of which are chemically equivalent. How many total signals would you expect in the 1 H NMR...
-
Predict the chemical shifts for the signals in the 1 H NMR spectrum of each of the following compounds: (a) (b) (c) (d) (e)
-
4. The following function creates a random number from 1 to 1000 (inclusive): import random def getRandom(): val = int(random.random() * 1000) + 1 return val a. Create a module with this single...
-
1. (5 marks) What should the pre-condition be in each of the following ten correctness statements for the statement to be an instance of Hoare's axiom scheme? All variables are of type int. (a) P { x...
-
Consider the following dataset. x1 x2 Class label -1 -1 -1 -1 0 4 4 2 1 -1 -2 0 2 -2 1 1 1 1 1 2 2 The given class label exhibits two natural clusters formed in the given dataset and acts as a ground...

Study smarter with the SolutionInn App


