Amino acids are the building blocks for all proteins in our bodies. A structure for the amino
Question:
Amino acids are the building blocks for all proteins in our bodies. A structure for the amino acid alanine is
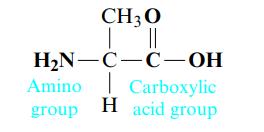
All amino acids have at least two functional groups with acidic or basic properties. In alanine, the carboxylic acid group has Ka = 4.5 × 10-3 and the amino group has Kb = 7.4 × 10-5. Because of the two groups with acidic or basic properties, three different charged ions of alanine are possible when alanine is dissolved in water. Which of these ions would predominate in a solution with [H+] = 1.0 M? In a solution with [OH-] = 1.0 M?
Transcribed Image Text:
CH 30 | || H₂N-C-C-OH Carboxylic Amino group H acid group
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 64% (14 reviews)
Solution a The ions include H OH and HCO The one w...View the full answer

Answered By

James Warinda
Hi! I’m James Otieno and I'm an experienced professional online tutor with countless hours of success in tutoring many subjects in different disciplines. Specifically, I have handled general management and general business as a tutor in Chegg, Help in Homework and Trans tutor accounts.
I believe that my experience has made me the perfect tutor for students of all ages, so I'm confident I can help you too with finding the solution to your problems. In addition, my approach is compatible with most educational methods and philosophies which means it will be easy for you to find a way in which we can work on things together. In addition, my long experience in the educational field has allowed me to develop a unique approach that is both productive and enjoyable.
I have tutored in course hero for quite some time and was among the top tutors awarded having high helpful rates and reviews. In addition, I have also been lucky enough to be nominated a finalist for the 2nd annual course hero award and the best tutor of the month in may 2022.
I will make sure that any student of yours will have an amazing time at learning with me, because I really care about helping people achieve their goals so if you don't have any worries or concerns whatsoever you should place your trust on me and let me help you get every single thing that you're looking for and more.
In my experience, I have observed that students tend to reach their potential in academics very easily when they are tutored by someone who is extremely dedicated to their academic career not just as a businessman but as a human being in general.
I have successfully tutored many students from different grades and from all sorts of backgrounds, so I'm confident I can help anyone find the solution to their problems and achieve
0.00
0 Reviews
10+ Question Solved
Related Book For 

Chemistry
ISBN: 9781305957404
10th Edition
Authors: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Question Posted:
Students also viewed these Engineering questions
-
All amino acids have at least two functional groups with acidic or basic properties. In alanine the carboxylic acid group has Ka = 4.5 10 -3 and the amino group has Kb = 7.4 10 -5 . Three ions of...
-
When acetone is dissolved in water containing 18O instead of ordinary 16O (i.e., H2 18O instead of H2 16O), the acetone soon begins to acquire 18O and becomes The formation of this oxygen-labeled...
-
From which of the standard amino acids are the following physiologically active amines derived? What modifications gave rise to these products? (a) GABA (b) Histamine (c) Thyroxine (d) Dopamine
-
Nisha has completed her MBA and has joined a company which was going to raise fund from long term sources such as Debt and Equity. Nisha was asked by her manager to prepare a report on which could be...
-
What is the principal regulatory authority when it comes to security offerings? What is its function?
-
Assign the following molecules to point groups: (a) HF, (b) IF7 (pentagonal bipyramid), (c) XeO2F2, (see-saw), (d) Fe,(CO)9 (22), (e) Cubane, C8H8, (f) Tetrafluorocubane, C8H4F4 (23) cO Fe co 23
-
A company that has 200 employees chooses a committee of 5 to represent employee retirement issues. When the committee is formed, none of the 56 minority employees are selected. (a) Use technology to...
-
Omega Chemicals Limited took a $ 420,000 two- year note receivable from a customer in connection with a major sale transaction on 1 May 20X7. The note required annual 31 April interest payments of...
-
How does catalytic dehydrogenation compare to hydrogenation in terms of mechanism, catalyst design, and application in industrial processes, such as the production of petrochemicals ?
-
Your organization is evaluating the quality of its call center operations. One of the most important metrics in a call center is Time in Queue (TiQ), which is the time a customer has to wait before...
-
Derive an equation analogous to the HendersonHasselbalch equation but relating pOH and pK b of a buffered solution composed of a weak base and its conjugate acid, such as NH 3 and NH 4 + .
-
A student dissolves 0.0100 mol of an unknown weak base in 100.0 mL water and titrates the solution with 0.100 M HNO 3 . After 40.0 mL of 0.100 M HNO 3 was added, the pH of the resulting solution was...
-
KopiPro is considering the purchase of a photocopying machine for $5,500 on December 31, 2016. It has a useful life of five years and a zero residual disposal price. Depreciation will be applied on a...
-
1.00 kilogram of liquid water at 50.0 C is converted completely to ice at -50.0 C . What is the change in entropy for the water. (Answer in kJ/K with correct sign)?
-
(1) Explain what is absolute advantage and what is comparative advantage? (2) Using the information given in the table to demonstrate the pattern of advantages between the two countries for the two...
-
a) Discuss the issue affecting the corporate workplace related to poor work-life balance. b) Identify, interpret, and diagnose appropriate organizational needs that require development and change...
-
Consider shares of common stock in Widgets, Inc. Suppose that the last dividend paid out to shareholders was $3.00 (Do = $3), the risk-free interest rate is 2%, and that the expected risk premium for...
-
Develop a minimum one-page report, written in first-person, past tense, chronological order that includes the following: Date, time, and location of the incident (use your creativity). The nature of...
-
Can a multithreaded solution using multiple user-level threads achieve better performance on amultiprocessor system than on a single-processor system?
-
Compare and contrast debt financing and equity financing as ways of starting a new business. Does one have an overall advantage over the other? What situation is more favorable to the use of debt...
-
What products would result after hydrolysis from reaction of the enamine prepared from cyclopentanone and pyrrolidine with the following , -unsaturated acceptors? (a) CH 2 =CHCO 2 Et (b) H 2 C=CHCHO...
-
Show how you might use an enamine reaction to prepare each of the followingcompounds: (b) (a) CH2CH2CO2CH3 CH2CH2CN
-
What product would you expect from a Robinson annulations reaction of 2-methyl-1, 3-cyclopentanedione with3-buten-2-one? -CH + H%3DH 2-Methyl-1,3-cyclo- pentanedione 3-Buten-2-one
-
Milligan's Backyard Storage Kits, a mail order company, sells a variety of backyard storage unit kits and landscaping decorations to its customers. Although the company makes a profit, David...
-
1. If you think about then Guest Services Essentials class, what do you believe to be the biggest change in your customer service practice? 2. Name and describe two customer service techniques to...
-
Packaging Waste & Recycling UK, 2013('000 tonnes) 2,260 Total Packaging Waste Total Recycled 4,000 3,868 3,459 3,000 2,339 2,000 1,639 1,029 1,000 806 714 462 436 23 0 0 Metal Glass Wood Paper...

Study smarter with the SolutionInn App


