Determine the phases present, the compositions of each phase, and the amount of each phase in wt%
Question:
Determine the phases present, the compositions of each phase, and the amount of each phase in wt% for the following Al2O3- Cr2O3 ceramics at 2150°C:
(a) Al2O3-30 wt% Cr2O3;
(b) Al2O3-50 wt% Cr2O3; and
(c) Al2O3-75 wt% Cr2O3.
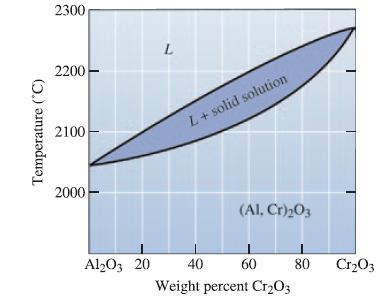
Transcribed Image Text:
Temperature (°C) 2300 2200 2100 2000 Al2O3 20 L L+ solid solution (Al, Cr)₂03 40 60 Weight percent Cr₂O3 80 Cr₂O3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 60% (10 reviews)
a Al2O330 wt Cr2O3 Phase 1 Liquid Al2O3Cr2O3 eutecti...View the full answer

Answered By

Hande Dereli
Enthusiastic tutor, skilled in ACT and SAT tutoring. Raised one student's score on the SATs from 1100 combined to 1400. Graduated with a 3.9 GPA from Davidson College and led a popular peer tutoring group for three years. Scored in the top 0.06% in the nation on the SATs. The real reason I'm the one to help you nail the test? Results. Clients invariably praise my ability to listen and communicate in a low-stress, fun way. I think it's that great interaction that lets me raise retest SAT scores an average of 300 points.
0.00
0 Reviews
10+ Question Solved
Related Book For 

The Science And Engineering Of Materials
ISBN: 9781305076761
7th Edition
Authors: Donald R. Askeland, Wendelin J. Wright
Question Posted:
Students also viewed these Engineering questions
-
Determine the phases that are present and the compositions for each phase in Cu-55 wt% Ag at 600C. Data Form Problem 11-23 Consider a Pb-70% Sn alloy. Determine (a) If the alloy is hypoeutectic or...
-
Determine the phases that are present and the compositions for each phase in Cu-85 wt% Ag at 800C. Temperature (C) 1200 1100 1000 900 800 700 600 500 400 300 200 0 10 20 30 40 50 60 70 80 90 100...
-
Al2O3 and Cr2O3 show 100% resolution among each other. a) Name the axes and regions by drawing a schematic phase diagram for the Al2O3-Cr2O3 system. (The melting temperatures of Al2O3 and Cr2O3 are...
-
Harry Bhel carries a business as a sole proprietorship. During its 2022 fiscal period, its first year of operations, the business had cash sales of $123,000. It also has sales on account of $46,000,...
-
Indicate the type of fiscal and monetary policies required to reach point F in the following figure (similar to Figure 18.10) for points C 3 , C 6 , C 9 , and C 12 .
-
The Cola Company reported the following comparative information at December 31 2020, and December 31, 2019 (amounts in millions and adapted): {Requirements} 1. Calculate the following ratios for 2020...
-
Refer to Exercise 9. Can you reject H0 at the = 0.01 level? State a conclusion. Exercise 9 Two suppliers of machine parts delivered large shipments. A simple random sample of 150 parts was chosen...
-
Starbucks is hoping to make use of its excess restaurant capacity in the evenings by experimenting with selling beer and wine. It speculates that the only additional costs are hiring more of the same...
-
Revenue and expense data for the current calendar year for Tannenhill Company and for the electronics industry are as follows. Tannenhill's data are expressed in dollars. The electronics industry...
-
The financial statements of Columbia Sportswear Company are presented in Appendix B. Financial statements of Under Armour, Inc. are presented in Appendix C. The complete annual reports, including the...
-
Determine the composition range in which the Tl-Pb alloy at 350C is (a) Fully liquid; (b) Fully solid; and (c) Partly liquid and partly solid. Refer to Figure 10-8(d) for the Tl-Pb phase diagram....
-
Determine the liquidus temperature, solidus temperature, and freezing range for the following NiO-MgO ceramic compositions: (a) NiO-30 mol% MgO; (b) NiO-45 mol% MgO; (c) NiO-60 mol% MgO; and (d)...
-
Create a process diagram in BPMN for the spa that shows activities from contacting Groupon for the first time through the end of the spas promotion.
-
The Poynting vector for an electromagnetic wave is given by \(\left(100 \mathrm{~W} / \mathrm{m}^{2} ight) \sin ^{2}\left[\left(1000 \mathrm{~m}^{-1} ight) z-\left(3.0 \times 10^{11} \mathrm{~s}^{-1}...
-
A parallel-plate capacitor has circular plates of radius \(R=\) \(0.10 \mathrm{~m}\) and a plate separation distance \(d=0.10 \mathrm{~mm}\). While a current charges the capacitor, the magnitude of...
-
At what speed does an electromagnetic wave pulse propagate through a dielectric for which the dielectric constant is \(\kappa\) ?
-
A force \(\vec{F}=F_{x} \hat{\imath}+F_{y} \hat{\jmath}\) with \(F_{x}=50 \mathrm{~N}\) and \(F_{y}=12 \mathrm{~N}\) is exerted on a particle as the particle moves along the \(x\) axis from \(x=1.0...
-
One thing that savvy growth-minded start-ups do is configure their products or services in ways that have built-in growth potential. Producing one-off products or services leads to limited growth...
-
The comparative financial statements for Prince Company are below: Required: 1. Complete the following columns for each item in the preceding comparative financial statements: INCREASE (DECREASE)...
-
Aztec Furnishings makes hand-crafted furniture for sale in its retail stores. The furniture maker has recently installed a new assembly process, including a new sander and polisher. With this new...
-
Although He is the second most abundant element in the universe, it is very rare on the earth. Why?
-
The xenon halides and oxides are isoelectronic with many other compounds and ions containing halogens. Give a molecule or ion in which iodine is the central atom that is isoelectronic with each of...
-
Hydrazine is somewhat toxic. Use the following half-reactions to explain why household bleach (highly alkaline solutions of sodium hypochlorite) should not be mixed with household ammonia or glass...
-
Employee longevity A large insurance company has developed a model to identify the factors associated with employee turnover. The dependent variable is number of years an employee stays with the...
-
5. Consider the 'top-hat' function which is zero everywhere except between -1 and 1 where it takes value 1. Show, using only elementary integration, that the convolution of this function with itself...
-
HERE IS THE SITUATION. With attendance plummeting by 50 percent in the past decade, a new, younger executive director was hired to reverse the trend. By understanding the importance of integrating...

Study smarter with the SolutionInn App


