Compare the structures of ethylene, acetylene, and benzene. Each of these compounds produces only one signal in
Question:
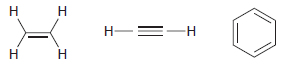
Transcribed Image Text:
Н Н Н- -н Н H.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 22% (9 reviews)
Increasing chemical shift in ...View the full answer

Answered By

Joram mutua
I am that writer who gives his best for my student/client. Anything i do, i give my best. I have tutored for the last five years and non of my student has ever failed, they all come back thanking me for the best grades. I have a degree in economics, but i have written academic papers for various disciplines due to top-notch research Skills.In additional, I am a professional copywriter and proofreader.
5.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Compare the structures of a synchondrosis and a symphysis.
-
Compare the structures of HNO2 and H2CO3. Which would you expect to be the stronger acid? Explain your choice.
-
Compare the structures of 1,4-pentadiene and divinyl amine: The first compound does not absorb UV light in the region between 200 and 400 nm. The second compound does absorb light above 200 nm. Using...
-
Starting with acetylene as your only source of carbon atoms, identify how you would prepare each member of the following homologous series of aldehydes: a. Ethanal b. Propanal c. Butanal d. Pentanal
-
What are three examples of popular messaging systems?
-
Nasreen Company issues 2 million, 10-year, 8% bonds at 97, with interest payable each January 1. a. Prepare the journal entry to record the sale of these bonds on January 1, 2025. b. Assuming instead...
-
Why does dynamic balancing imply static balancing?
-
Comprehensive problem on unit costs, product costs Tampa Office Equipment manufactures and sells metal shelving. It began operations on January 1, 2009. Costs incurred for 2009 are as follows (V...
-
Joe aged 24, and Kate, aged 24, met in college. While studying, Joe worked part-time selling car parts to autobody shops in the GTA. He earned $12,000 a year, for two years. After graduating from...
-
Certain item descriptions and amounts are missing from the monthly schedule of cost of goods manufactured and income statement of Charlie Manufacturing Company. Fill in the blanks with the missing...
-
How many signals will be expected in the 1 H NMR spectrum of each of the following compounds? (a) (b) (c) (d) (e) (f) (g) (h) `NO2
-
The 2009 balance sheet and income statement for the Woods Company are shown here: Woods Company: Income Statement for the Year Ending December 31, 2009 ($ thousands)...
-
Complete the following. (a) State the degree and leading coefficient of f. (b) State the end behavior of the graph of f. f(x) = x - 4x + 3x - 3
-
A warehouse of 6,000 square feet stocks three items. Using the item data below, find the appropriate order quanti- ties. Random access storage is used so that space planning can be based on average...
-
Explain the differences between capital expenditure and capital budgeting. Provide examples for each within the context of a hospital environment. Discuss how they relate to each othe?
-
Suppose on January 1 the value of the euro is at $1.17/euro, and later in March 1 the value of is at $1.03/Euro. By how much did the dollar appreciate or depreciate? Enter your response in percentage...
-
If i have a savings account investment that has an expected return of $127628 and an inflation rate of 2.1% how would i calculate the amount of money the return turns into with the cost of inflation...
-
Yellow Shoe Company, a distributor, purchases all its shoe inventory from Styles Limited. Selected account balances are included in the following table: Dec. 31, Year 1Dec. 31, Year...
-
Simplify the expression. (20m + 8y 32z)
-
Do public and private companies follow the same set of accounting rules? Explain.
-
How would you carry out the following transformations? More than one step is required in each case. (a) H (b) Ph "Co "
-
The following pKa values have been measured. Explain why a hydroxyl group in the para position decreases the acidity while a hydroxyl group in the Meta position increases the acidity. . CO2H CO2H...
-
3-Methyl-2-hexenoic acid (mixture of E and Z isomers) has been identified as the substance responsible for the odor of human sweat. Synthesize the compound from starting materials having five or...
-
write a page on global migration giving statistics and then focus on migration within Africa and then South Africa. Write reference?
-
Lesotho'footwear industry manufacturer one in maseru and other in maputsoe.aputsoe.in maseru te marginal benefit associated wih ppolluiio cleeanup is MB = 3 0 0 - 1 0 Q while in maputsoe te marginal...
-
Under what conditions would the Fed choose to decrease the money supply, how would it do so , and what is the goal of doing so ? How does the Fed factor inflation into its actions?

Study smarter with the SolutionInn App


