Consider the following reaction: a) How would the rate be affected if the concentration of tertbutyl bromide
Question:
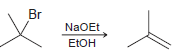
a) How would the rate be affected if the concentration of tertbutyl bromide is doubled?
b) How would the rate be affected if the concentration of sodium ethoxide is doubled?
Transcribed Image Text:
Br NaOEt ETOH
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 80% (10 reviews)
a The rate of an E2 process is dependent on the concentrations of the s...View the full answer

Answered By

GERALD KAMAU
non-plagiarism work, timely work and A++ work
4.40+
6+ Reviews
11+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Consider the following reaction at some temperature: H2O(g) + CO(g) H2(g) + CO2(g) K = 2.0 Some molecules of H2O and CO are placed in a 1.0- L container as shown below. When equilibrium is reached,...
-
Consider the following reaction at equilibrium: From the data shown here, calculate the equilibrium constant (both KP and Kc) at each temperature. Is the reaction endothermic or exothermic?...
-
Consider the following reaction: CH3X + Y CH3Y + X At 25oC the following two experiments were run, yielding the following data: Experiment 1: [Y]0 = 3.0 M Experiment 2: [Y] 0 = 4.5 M Experiments were...
-
Villaverde Company insures the life of its president for P8,000,000, the corporation being the beneficiary of an ordinary life policy. The premium is P200,000. The policy is dated January 1, 2010....
-
Company to be analyzed: Delta Airlines, Inc, and report structure and requirements are: a. An analysis overview which includes background information of the company and the industry. This should...
-
A spherical balloon filled with helium gas must lift a \(1.50-\mathrm{kg}\) scientific payload off the ground, and the mass of the empty balloon is \(0.500 \mathrm{~kg}\). (a) What is the minimum...
-
True or False. A singular point corresponds to a state of equilibrium of the system.
-
Celtex is a large and very successful decentralized specialty chemical producer organized into five independent investment centers. Each of the five investment centers is free to buy products either...
-
Southwest Milling Company purchased a front-end loader to move stacks of lumber. The loader had a list price of $122,860. The seller agreed to allow a 4.25 percent discount because Southwest Milling...
-
American International Automotive Industries (AIAI) manufactures auto and truck engine, transmission, and chassis parts for manufacturers and repair companies in the United States, South America,...
-
In the algebraic version of prospect theory, the variable x represents gains and losses. A positive value for x is a gain, a negative value for x is a loss, and a zero value for x represents...
-
Ted has always had difficulty saving money. So on June 1 st , Ted enrolls in a Christmas savings program at his local bank and deposits $750. That money is totally locked away until December 1 st so...
-
Use f(x) and g(x) to find a formula for each expression. Identify its domain. (a) (f + g)(x) (c) (fg)(x) (b) (f- g)(x) (d) (f/g)(x)
-
What theory best explains why Chris's brother became so upset when people called Chris a junkie?
-
How can World Taekwondo be useful because, with the recent rise of awareness of bullying in school and online, parents are turning to martial arts to help kids improve their self-confidence and learn...
-
Simplify the following complex numbers into the standard polar form (i.e., z=ree with r0 and - <0): (a) z = 6+3 (b) 22 = 2+47 23 = je/4 (c) 23 =
-
5. Simplify the following expressions into both rectangular form and standard polar form (reje with r0 and - <0). (a) 4e-j/3-3e-j*/6 (b) (-2+ j2)10 (c) R{(1+j)e-j/4} (d) (2-j2)1/3
-
Consider the contrasting physical development between the genders at the beginning of adolescence, especially for the boys. Think also of the exponential growth made in the following years. What does...
-
Evaluate the binomial coefficient. 4 C 0
-
What can scientists learn by comparing the fossilized skeletons of extinct primates with the bones of modern species?
-
How might the following compounds be prepared using Michael reactions? Show (he nucleophilic donor and the electrophilic acceptor in eachcase. ", , (b) . (a) CHCCH-CH2CH2CH3 CH2H2CCgHs o2Et NO2 (d)...
-
The so-called Wieland?Miescher ketone is a valuable starting material used in the synthesis of steroid hormones. How might you prepare it from 1, 3-cyclohexanedione? Wieland-Miescher ketone
-
The following reactions are unlikely to provide the indicated product in high yield. What is wrong witheach? . , (a) Na* "OEt CHH2CH CHCH CHCH Ethanol (b) .oon CH2CH2CCH3 Na* "OEt + H3 Ethanol "H...
-
Give a summary of concepts and the applications for the following quistion: When a 200.0-g mass is attached to a spring, it stretches the spring by 7.50 cm. With that mass-spring system in...
-
A potato is launched out of a building (building A) from the 10th floor which is 15 m high, with a velocity of 55 m/s at an angle of 35.0 above the horizontal. There is a building (building 2) 130 m...
-
An eagle is flying horizontally at a speed of 2.9 m/s when the fish in her talons wiggles loose and falls into the lake 4.4 m below. Calculate the magnitude of the velocity of the fish relative to...

Study smarter with the SolutionInn App


