Tell the number of hydrogens bonded to each carbon atom in the following substances, and give the
Question:
Tell the number of hydrogens bonded to each carbon atom in the following substances, and give the molecular formula ofeach:
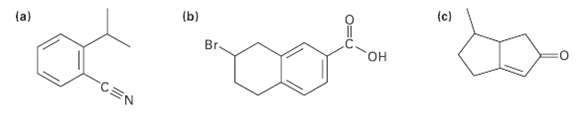
Transcribed Image Text:
(c) (b) (a) Br. он CEN
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 60% (10 reviews)
a 1 H 1 H 1 H 1H 1 OH 10...View the full answer

Answered By

Akhilandeswari Thiruneelakandan
I'm a mechanical engineer by profession and math enthusiast by passion. I love solving complex problems and I tutor in various online platforms like studypool and study.com. I am also an academic expert volunteer with an NGO Make A Difference, wherein I tutor 11th and 12th grade kids at a shelter home.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Tell how much hydrogen is bonded to each carbon in the following compounds, and give the molecular formula of each substance: (b) NHCH3 Adrenaline Estrone (a hormone)
-
Star (*) each asymmetric carbon atom in the following examples, and determine whether it has the (R) or (S) configuration. (a) (b) (c) (d) (e) (f) (g) (h) (i) CH H CH,CH, H,C CH,CH, H H CH CCH, CI H...
-
Indicate the molecular geometry around each carbon atom in the compound H;CCH=CHCH,CCH,COOH
-
Draw a graph showing the marginal revenue (MR) and marginal cost (MC) of increasing the tax rate (t) in the Peltzman model of regulation represented by equation (19.1). Place the tax rate on the...
-
Opal, Inc., is about to make its first attempt to borrow from a local bank. The company uses LIFO for tax purposes, solely to defer taxes, and believes that income computed using the FIFO method...
-
The density of 70.5 wt% aqueous perchloric acid is 1.67 g/mL. Recall that grams refers to grams of solution ( = g HClO4 + gH2O). (a) How many grams of solution are in 1.000 L? (b) How many grams of...
-
An aircraft cruises at a Mach number of 2.0 at an altitude of \(15 \mathrm{~km}\). Inlet air is decelerated to a Mach number of 0.4 at the engine compressor inlet. A normal shock occurs in the inlet...
-
Following the 1990 Iraqi invasion of Kuwait, the price of crude oil soared, as did retail gasoline prices. This led the major U. S. oil companies to try to hold down their reported earnings. The oil...
-
Explain the significance of multi-scale modeling in process simulation. How does the integration of different scales, from molecular to plant-level simulations, provide a comprehensive view of...
-
Rainbow Life Insurance Company, based in Toronto, uses a job evaluation system of their own design. The system is based on five job characteristics: Skill, Mental Effort, Physical Effort,...
-
Convert the following structures into skeletal drawings: (a) (b) H. H. H. Indole 1,3-Pentadiene (d) (c) H. -CI C-CI H. 1,2-Dichlorocyclopentane Benzoquinone U
-
Propose structures for molecules that meet the following descriptions: (a) Contains two sp2-hybridized carbons and two sp3-hybrized carbons (b) Contains only four carbons, all of which are...
-
In Problems 95106, f () = sin and g() = cos. Find the exact value of each function below if = 60. Do not use a calculator. (7)
-
The graph of a function is given. Use the graph to estimate the following. (Enter your answers using interval notation.) 4 (a) The domain and range of f domain
-
I.Trend Analysis : Table 1 [1]: 2020 2019 2018 2017 2016 Sale Growth -2.71% 0.77% 3.83% 0.04% -4.29% COGS Growth -4.88% -1.65% 5.98% 1.72% Gross Income Growth -0.15% 3.79% 1.27% -1.88% Interest...
-
Coat color in mice is controlled by two genes. The M/m gene encodes an enzyme that makes a black pigment while A/a makes yellow pigment. The dominant M allele results in normal black pigment...
-
Determine The Better Of The Two Alternatives Using The Present Worth Analysis. Use An Interest Rate Of 7%. 1. Initial Cost Annual Benefit Salvage Value Life In Years MARR It. X 12,500 6,800 55,000...
-
Using the production possibility frontier and social indifference curve that represents social preferences, explain how trade can lead to greater social welfare. Use equations and graphs to explain....
-
Based on Exhibit 1, the manager could have delivered more value to the portfolio during the investment period by weighting more toward: A. value stocks. B. small-cap stocks. C. momentum stocks....
-
Separate variables and use partial fractions to solve the initial value problems in Problems 18. Use either the exact solution or a computer-generated slope field to sketch the graphs of several...
-
A fluid obeys the equation of state a. For what values of the constants B and C will this fluid undergo a vapor-liquid phase transition? b. What is the molar internal energy change if this fluid is...
-
What is the hybridization at the N and each C in this molecule? Indicate the type of bond and the orbitals that are overlapping to form it for each of the designated bonds (for example, ?CSP3 + H1s)....
-
Consider hydrogen cyanide, H C N. (a) What is the hybridization at the N at the C? (b) What are the types of the three CN bonds? What orbitals overlapping to form them? (c) In what type of orbital...
-
What is the hybridization at each C in this molecule? Indicate the type of bond and the orbital's that are overlapping to form it for each of the designated bonds? ITT H=C=C=C=C_C7H (both) H tall...
-
Assume you have a portfolio consisting of a $400,000 investment in stock A and a $600,000 investment in stock B. Suppose that the daily volatilities of these two assets are 2% and 1.5%, respectively,...
-
What is the role of education in promoting social mobility and reducing inequality, and how can educational systems be reformed to better serve diverse populations while preparing individuals for the...
-
A European call option and put option on a non-dividend paying stock both have a strike price of $51 and an expiration date in 6 months. The put sells for $2.50 and the call sells for $2. The...

Study smarter with the SolutionInn App


