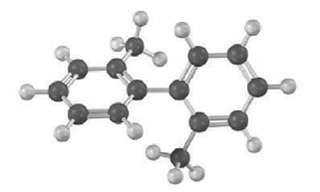
The following molecular model of a dimethyl-substituted biphenyl represents the lowest-energy conformation of the molecule. Why are
Question:
The following molecular model of a dimethyl-substituted biphenyl represents the lowest-energy conformation of the molecule. Why are the two benzene rings tilted at a 63o angle to each other rather than being in the same plane so that their p orbital?s can overlap? Why doesn?t complete rotation around the single bond joining the two rings occur
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 58% (12 reviews)
In the lowestenergy conformation of the biphenyl the aromatic rings are ...View the full answer

Answered By

Muhammad Umair
I have done job as Embedded System Engineer for just four months but after it i have decided to open my own lab and to work on projects that i can launch my own product in market. I work on different softwares like Proteus, Mikroc to program Embedded Systems. My basic work is on Embedded Systems. I have skills in Autocad, Proteus, C++, C programming and i love to share these skills to other to enhance my knowledge too.
3.50+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
The following molecular model represents a tetrahedral intermediate resulting from addition of a nucleophile to an aldehyde or ketone. Identify the reactants, and write the structure of the final...
-
The following molecular model is a representation of para-aminobenzoic acid (PABA), the active ingredient in many sunscreens. Indicate the positions of the multiple bonds. And draw a skeletal...
-
The following molecular model is that o a carbocation. Draw two resonance structures for the carbocation, indicating the positions of the doublebonds.
-
Suppose school records reveal that historically, 10 % of the students in Milton High School have dropped out of school. What is the probability that more than two students in a class of 30 will drop...
-
What are the guidelines regarding the deductibility of luxury water travel (e.g., cruise ships) for business purposes? IRS Publication 463.
-
Measure the width at half-height of the tallest peak in the spectrum below and calculate the resolving power of the spectrometer from the expression m/m 1/2 . Would you expect to be able to...
-
P(x > 170) The random variable x is normally distributed with mean = 174 and standard deviation = 20. Find the indicated probability.
-
The ABC Corporation is considering a joint venture with another company to start an Internet based business. The new business will tap into the expertise ABC has developed in writing the complex...
-
An investor purchased $100,000 in a 5-year index-linked guaranteed investment certificate (GIC) in a non-registered account. The GIC matured at $110,000. Assuming he is in a 26% marginal tax bracket,...
-
For one of the binary systems listed in Table 13.10, based on Eq. (13.19) and the Wilson equation, make the following calculations: (a) BUBL P : t = 60C, x 1 = 0.3. (b) DEW P : t = 60C, y 1 = 0.3....
-
Draw the product from reaction of each of the following substances with (i) Br2, FeBr3 and (ii) CH3COCl,AlCl3. (b) (a)
-
How would you synthesize the following compound starting from benzene? More than one step isneeded.
-
How much money would have to be deposited today to accumulate a. $10,000 after 6 years if the investment earns 5%/year compounded annually. b. $6,500 after 4 years if the investment earns 8...
-
Prove that, (i) sinh = ln(2 + z + 1) (ii) coth- = = 1/ h ( + ) In 2 -
-
Do stock prices reflect/embed available information (eg., the economic impact of the Pandemic)?. Argue if the markets are efficient or the markets are inefficient.
-
As data flows through a simple accounting system it proceeds from source documents to the general journal to the general ledger, and to the worksheet. What is the final location to which the data...
-
What should you consider when preparing the load/client?
-
Explain Cost allocation is a critical part of the costing process because it addresses the issue of how to assign the costs of support activities to the revenue-producing (patient services) model....
-
The international strategy suggested by McMahon is most likely characterized as: A. risk based. B. return oriented. C. diversification oriented. The Mackenzie Education Foundation funds educational...
-
Uniform electric field in Figure a uniform electric field is directed out of the page within a circular region of radius R = 3.00 cm. The magnitude of the electric field is given by E = (4.50 x 10-3...
-
Evaluate the difference for the ideal and van der Waals gases, and for a gas that obeys the virial equation of state. (3) - (0) P V
-
Show the products of thesereactions: Br ELOH a) CH3CH,CH + CH;CH,0 I 1) Na 2) CH,I CH,CH,CH,CH, NaOH b) c) ELOH CH3
-
Diphenhydramine can also be synthesized by heating bromo diphenyl methane with the amino alcohol shown here. Offer a reason why the oxygen, rather than the nitrogen, of this compound acts as the...
-
Explain which route would provide a better synthesis of theseethers: CH3 CH, CH3 CHI + CH,CO a) CH.O + CH,CCI CH3 CH,COCH3 H3 CH3 CH, CH,OCHCH,CH; CH,Br CH,0 Br + CH;CHCH,CH3 b) + CH;CHCH,CH,
-
Popular furniture company, IKEA, has purchased forests in Romania as well as land in Alabama to assist with keeping up with the wood demand necessary to complete customer orders. This was one way...
-
How does China being Turkey's biggest import partner affect Turkey's exchange rate?
-
Assignment 4 In this assignment you are provided information on an experiment and you are required to investigate and interpret the output which is provided below. Problem: Consider the...

Study smarter with the SolutionInn App


