2-Brorno-6, 6-dimethylcyclohexanone gives 2, 2-dirnethylcyclopentane- carboxylic acid on treatment with aqueous NaOH followed by acidification, a process
Question:
2-Brorno-6, 6-dimethylcyclohexanone gives 2, 2-dirnethylcyclopentane- carboxylic acid on treatment with aqueous NaOH followed by acidification, a process called the Favorskii reaction. Propose a mechanism
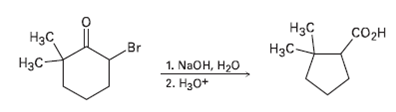
Transcribed Image Text:
Нзс Нас Соон Br 1. NaOH, H20 2. На0+ Нас H3C-
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (16 reviews)
H3C H3C 0 OH ...View the full answer

Answered By

Chiranjib Thakur
I have no tutoring experience yet, but I can share my skills and knowledge gained from my education and work experiences. I have been a CPA since 2012 with 6 years of work experience in internal auditing and 4 years of work experience in accounting at the supervisory level.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Propose a mechanism for the reaction of benzoic acid with oxalyl chloride. This mechanism begins like the thionyl chloride reaction, to give a reactive mixed anhydride. Nucleophilic acyl substitution...
-
A compound reacts with methylmagnesium bromide followed by acidification to form the product with the following 1H NMR spectrum. Identify the compound. 6 (ppm) 2 frequency
-
A carboxylic acid has two oxygen atoms, each with two nonbonding pairs of electrons. (a) Draw the resonance forms of a carboxylic acid that is protonated on the hydroxyl oxygen atom. (b) Compare the...
-
Write the Comparison for Legal Factors of China and South Africa
-
Assume that a manager becomes the leader of a division in which the vast majority of the workers are under 25, such as a restaurant chain. Would you recommend that the leader get some body piercing...
-
G. Menges developed the following econometric model for the West German economy: where Y = national income I = net capital formation C = personal consumption Q = profits P = cost of living index R =...
-
Consider the flow past a flat plate. Here we examine the effect of a superimposed velocity \(v_{y 0}\) at the plate surface. This can be done by the integral method with only minor changes in the...
-
Create an enterprise data model that captures the data needs of FAME. Use a notation similar to the one shown in Figure 1-4. In figure 1-4 INVENTORY PRICING HISTORY CUSTOMER Places Keeps Price...
-
he treasurer at Dell estimated the expected return on equity for the firm is 14.79%. The current T-bill rate is 5.90%, the expected market return is 16.61% and the market premium is 10.71%. What is...
-
On January 1, 2014, Prince Corporation acquired 70% of the 100,000 outstanding voting shares of Song Limited for a cash consideration of $1,015,000. On that date, shares of Song Limited were trading...
-
Identify the missing reagents af in the following scheme: Br "o2 "
-
In plants, terpenes are biosynthesized by a pathway that involves loss of CO2 from 3-phosphornevalonate 5-diphosphate to yield isopentenyl diphosphate. Use curved arrows to show the mechanism of this...
-
Business week magazine currently offers the following subscription options: one year for $39; two years for $72; three years for $103. These rates are expected to increase at the general inflation...
-
Within six months, a 35% rise in revenue is part of the aim. Which three important business-related topics should the company plan emphasise?
-
The effective interest rate on bonds payable reflects the effective cost of borrowing at what interest rate?
-
Which of the Contributions to a Traditional IRA or a Traditional 401(k) plan is an example of what type of income? Explain.
-
For 2022, Rebecca, age 52, has $5,972 of net self-employment income from her pet grooming business. Her self-employment tax is $844. What is the maximum amount that Rebecca may contribute to a...
-
A department submitted a request for an increase in the budget. The following justification was provided The department currently has a budget of $2,684,568. The current expenses for services are...
-
Let \(X\) be a single observation from a discrete distribution with probability distribution function \[f(x \mid \theta)= \begin{cases}\frac{1}{4} \theta & x \in\{-2,-1,1,2\} \\ 1-\theta & x=0 \\ 0 &...
-
Design and describe an application-level protocol to be used between an automatic teller machine and a bank's centralized computer. Your protocol should allow a user 's card and password to be...
-
Palladium crystallizes with a face-centered cubic structure. It has a density of 12.0 g/cm 3 , a radius of 138 pm, and a molar mass of 106.42 g/mol. Use these data to calculate Avogadros number.
-
Draw a lewis structure for each of the following species. Show all unshared pairs and the formal charges, if any. Assume that bonding follows the octet rule in all cases. (a) NH3 ammonia (b) [NH4]+...
-
Predict the approximate bond angles in each of the following molecules. (a) BeH2 (b) (c) :Cl Si HCC CH (Give H C C and C) _C-C angles. allene
-
The percent s character describes the hybridization of an orbital. For example, an ,sp3 orbital has 25%s character. Given the bond angles in each case, calculate the percent s character of (1) the...
-
1. Refer to the graph provided. Price, cost of unit $15- 9 MC ATC MR = P = D a. At what level of output does the firm maximize profit? Explain how you know. b. At the profit-maximizing quantity of...
-
A bond issued 10 years ago had a face value of $2,000; a coupon rate of 5%; and a yield of 6% when it was sold last month in the secondary bond market. At what price did the bond sell in the...
-
What are the assertions affected by the earlier list on what could go wrong in the post to the general journal process? The assertions to use are Completeness Existence/Occurrence Presentation and...

Study smarter with the SolutionInn App


