Question: A bubble-cap tray absorber is designed to absorb 40% of the propane from a rich gas at 4 atm. The specifications for the entering rich
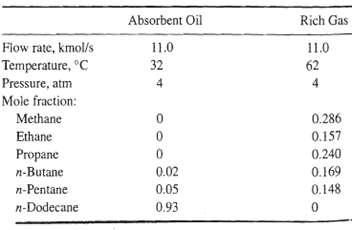
A bubble-cap tray absorber is designed to absorb 40% of the propane from a rich gas at 4 atm. The specifications for the entering rich gas and absorbent oil are as follows:
(a) Determine the number of equilibrium stages required and the splits of all components.
(b) Determine the actual number of trays required and the splits and Murphree vapor-tray efficiencies of all components.
(c) Compare and discuss the equilibrium-based and rate-based results. What do youconclude?
Absorbent Oil Rich Gas Fiow rate, kmol/s 11.0 11.0 Temperature, C 32 62 Pressure, atm Mole fraction: Methane 0.286 Ethane 0.157 0.240 Propane 0.02 n-Butane 0.169 0.148 n-Pentane 0.05 n-Dodecane 0.93
Step by Step Solution
3.40 Rating (181 Votes )
There are 3 Steps involved in it
The ChemSep program was applied to both cases using the SRK method for Kvalues and enthalpies a It was determined for the equilibrium stage method that the average Kvalue for propane was about 30 Ther... View full answer

Get step-by-step solutions from verified subject matter experts
Document Format (1 attachment)
37-E-C-E-S-P (451).docx
120 KBs Word File


