Question: A process instrument reading, Z (volts), is thought to be related to a process stream flow rate V(L/s) and pressure P(kPa) by the following expression:
A process instrument reading, Z (volts), is thought to be related to a process stream flow rate V(L/s) and pressure P(kPa) by the following expression:
Process data have been obtained in two sets of runs-one with V held constant, the other with P held constant. The data are as follows:
(a) Suppose you had only performed runs 2, 3 and 5. Calculate a, b, and c algebraically from the data for these three runs.
(b) Now use a graphical method and all the data to calculate a, b, and c. Comment on why your would have more confidence in this result than in that of part(a).
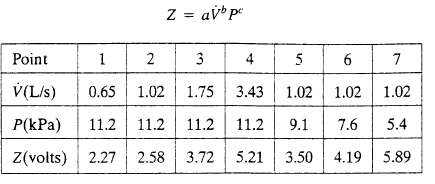
Z = avb pe Point 1 2 3 4 5 V(L/s) 0.65 1.02 1.75 3.43 1.02 1.02 1.02 P(kPa) 11.2 11.2 11.2 11.2 9.1 7.6 5.4 Z(volts) 2.27 2.58 3.72 5.21 3.50 4.19 5.89 6 7
Step by Step Solution
3.35 Rating (164 Votes )
There are 3 Steps involved in it
a For runs 2 3 and 4 Z av plnZ lna blnclnp In35 Ina b ln102cln91 In25... View full answer

Get step-by-step solutions from verified subject matter experts
Document Format (2 attachments)
13-E-C-E-C-P (38).pdf
180 KBs PDF File
13-E-C-E-C-P (38).docx
120 KBs Word File


