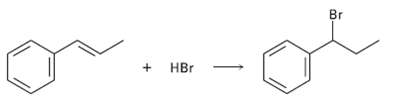
Addition of HBr to 1-phenyipropene yields only (1-bromopropyl) benzene. Propose a mechanism for the reaction, and explain
Question:
Addition of HBr to 1-phenyipropene yields only (1-bromopropyl) benzene. Propose a mechanism for the reaction, and explain why none of the other regioisomer isproduced.
Transcribed Image Text:
Br + HBr
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 53% (13 reviews)
HBr o Protonation of the double bond ...View the full answer

Answered By

Joseph Mwaura
I have been teaching college students in various subjects for 9 years now. Besides, I have been tutoring online with several tutoring companies from 2010 to date. The 9 years of experience as a tutor has enabled me to develop multiple tutoring skills and see thousands of students excel in their education and in life after school which gives me much pleasure. I have assisted students in essay writing and in doing academic research and this has helped me be well versed with the various writing styles such as APA, MLA, Chicago/ Turabian, Harvard. I am always ready to handle work at any hour and in any way as students specify. In my tutoring journey, excellence has always been my guiding standard.
4.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Propose a mechanism for the reaction of acetyl chloride with phenylmagnesium bromide to give 1,1-diphenylethanol. OH (1) ether solvent CH3-C CI 2 (2) H 0 acetyl chloride phenylmagnesium bromide...
-
Propose a mechanism for the reaction of cyclohexyl methyl ketone with excess bromine in the presence of sodium hydroxide.
-
Propose a mechanism for the reaction of an amide with thionyl chloride to form a nitrile.
-
How did your parents communication with you influence your self-concept?
-
Sam, age 45, is single. For 2016, he has the following items: Business income........................................................$70,000 Business...
-
1. Some criticize the actions of companies that try to align themselves with the values and views of subcultures as simply moves to make more money rather than attempting to make a social statement....
-
The Student Winner was Ulf Aslak Jensen, for the video "How People Gather: An Interactive Visualization Approach." Watch this video, and answer the following questions: (a) What data are this video...
-
Fun Ltd., a Texas company, is an expert in the assembly of a variety of video games consoles and they also offer repair parts for these systems. The products range from small handheld consoles that...
-
The demand for the new Christian Paboutin shoes can be written as a function of its selling team of the shoe company estimates that there is a base demand for 430000 shoes (mean will buy the shoes if...
-
Ceramic Structures has experienced rapid growth over the past several years. Sales are expected to grow at 15% per year for the next three years. Sales growth has been fueled by aggressive pricing as...
-
Electrophilic substitution on 3-phenylpropanenitrile occurs at the ortho and Para positions, but reaction with 3-phenylpropenenitrile occurs at the meta position. Explain using resonance structures...
-
Triphenylmethane can be prepared by reaction of benzene and chloroform in the presence of A1Cl3. Propose a mechanism for thereaction. H. + CHCI3 AICI3
-
2-Iodobenzoic acid, a tan, crystalline solid, can be prepared from 2-aminobenzoic acid. Other required reagents are NaNO 2 and KI (as well as HCl). (a) If you use 4.0 g of 2-aminobenzoic acid, 2.2 g...
-
Suppose a dentist is organized as a sole proprietorship. Last year, the dentist's total revenue was $320,000 and total costs were $250,000. The dentist left a job paying $112,00 a year to start the...
-
If inflation in the US is higher than in Japan, what will happen to the exchange rate between the US dollar and the Japanese yen?
-
According to Groom and Nass, why should robots be a source of moral concern? What are the main considerations for rethinking how robots can help humans, in your reasoned opinion? Evaluate and answer,...
-
in an opinion column on Wall Street diary, Princeton University economist Alan Blinder asks, "Is the Phillips curve dead?" Source: Alan S. Blinder, "Is the Phillips Curve Dead? And Other Questions...
-
Recall the 2009 Stimulus Package Success Application to answer the following question(s). Obama's stimulus package was implemented to help the economy recover from the recession. This package was...
-
How did the Jacksonville, Florida, vagrancy ordinance invalidated by the Supreme Court in the Papachristou case offend the Constitution of the United States? Have the reforms in vagrancy laws at the...
-
(a) Given a mean free path = 0.4 nm and a mean speed vav = 1.17 105 m/s for the current flow in copper at a temperature of 300 K, calculate the classical value for the resistivity of copper. (b)...
-
Derive the following Maxwell relations for open systems. a. Starting from Eq. 6.2-5a, b. Starting from Eq. 6.2-6a, c. Starting from Eq. 6.2-7a, d. Starting from Eq. 6.2-8a, (). HT ON (0) aN S, N S,V...
-
Show the products of thesereactions: CH NaOH Br2 Bra b) a) . .
-
The reaction of an alkenes with bromine in an alcohol as solvent produces as ether as the product. Show a mechanism for the following reaction and explain the stereochemistry of theproduct. Br . H....
-
Show all the steps in the mechanism for the formation of MTBE from methanol and isobutylene.
-
Assume that a robot systems constant failure and repair rates are 0.0004 failures per hour and 0.002 repairs per hour, respectively. Calculate the robot system steady-state unavailability and...
-
March 6, 2018 David Beckham's Latest Pitch: Men's Cosmetics. By Robert Williams Men of the world, David Beckham has a message for you: Moisturize. "It's the biggest thing," he says in a London...
-
3. On January 1, an investment fund was opened with an initial balance of 5000. Just after the balance grew to 5200 on July 1, an additional 2600 was deposited. The annual effective yield rate for...

Study smarter with the SolutionInn App


