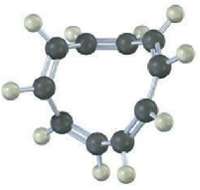
Question: A All-cis Cyclodecapentaene is a stable molecule that shows a single absorption in its 1H NMR spectrum at 5.67 ?. Tell whether it is aromatic,
A All-cis Cyclodecapentaene is a stable molecule that shows a single absorption in its 1H NMR spectrum at 5.67 ?. Tell whether it is aromatic, and explain its NMR spectrum.
Step by Step Solution
3.23 Rating (167 Votes )
There are 3 Steps involved in it
The allcis Cyclodecapentaene shown here is not aromatic be... View full answer

Get step-by-step solutions from verified subject matter experts
Document Format (1 attachment)
22-C-O-B (89).docx
120 KBs Word File


