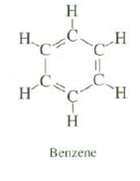
Although carbon?carbon double bonds are shorter than carbon?carbon single bonds, all of the carbon?carbon bonds of benzene
Question:
Although carbon?carbon double bonds are shorter than carbon?carbon single bonds, all of the carbon?carbon bonds of benzene are the same length. Explain.
Transcribed Image Text:
H. H C. H H Benzene H H
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 72% (11 reviews)
The actual structure of benzene is a h...View the full answer

Answered By

Stacy kosgei
I offer quality, original and timely services; Highly credible and void of plagiarism. Your success is my pleasure.
5.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
The hydrocarbons acetylene (C2H2) and benzene (C6H6) have the same empirical formula. Benzene is an "aromatic" hydrocarbon, one that is unusually stable because of its structure. (a) By using the...
-
Bonds between carbon and oxygen (C O) are more polar than bonds between sulfur and oxygen (S O). Nevertheless, sulfur dioxide (SO 2 ) exhibits a dipole moment while carbon dioxide (CO 2 ) does not....
-
Although perpetual bonds are illegal in the United States, sometimes it is easiest to assume that interest payments last forever to show some simple results based on Equation. Use that equation to...
-
If we take $1 away from a rich person and give it to a poor person, the rich person loses less utility than the poor person gains. Comment.
-
Visit the website of a major airline to find information about the airline's mission, objectives, strategic priorities, organizational structure and operations. Required: 1. Outline the airline's...
-
Classify each of the data elements in the Sales Transactions database (Figure 1.1) as categorical, ordinal, interval, or ratio data and explain why. Figure 1.1 A B 1 Sales Transactions: July 14 E H....
-
Asphalt at \(120^{\circ} \mathrm{F}\), considered to be a Newtonian fluid with a viscosity 80,000 times that of water and a specific gravity of 1.09, flows through a pipe of diameter \(2.0...
-
On May 31, 2012, Laban Company had a cash balance per books of $5,681.50. The bank statement from Citizens Bank on that date showed a balance of $7,964.60. A comparison of the statement with the Cash...
-
The wind power density is the power in the wind per unit area. Using your learning resources, calculated the wind power density given an air density of 1.16 kg/m 3 and a windspeed of 3.9 m/s,
-
Read the given description of a CSP and write solution as per the given questions: [20] YCPS@GIFT is going to arrange a gaming competition at campus. They are considering using seven games for this...
-
Chlorine is more electronegative than phosphorus. Predict the dipole moment of PCl5.
-
Explain whether or not these molecules are polar (have a dipole moment). (a) CBr 4 (b) NH 3 (c) CH 3 OCH 3 (d) CH 2 C1 2 (e) CO 2
-
Phoenix Corp. purchased Machine no. 201 on May 1, 2014. The following information relating to Machine no. 201 was gathered at the end of May: Price ....................... $85,000 Credit terms...
-
What is dividend?
-
Do you think it is possible to have win-win negotiations? Why or why not?
-
Differentiate between Type I and Type II errors.
-
Briefly describe the test statistic.
-
Consider a market with two securities, one of them risk free with rate of return \(R\), and three future states. Suppose that the risky security has a present value \(S_{1}\) and can take three...
-
Elemental copper, Cu, is copper colored. Elemental sulfur, S8, is yellow. What does this tell you about the color of the compound copper sulfide, CuS?
-
CRUZ, INC. Comparative Balance Sheets December 31, 2015 CRUZ, INC. Income Statement For Year Ended December 31, 2015 Required Use the indirect method to prepare the cash provided or used from...
-
Determine an appropriate viewing rectangle for the function f(x) = 8 2x 2 and use it to graph f.
-
How many products might be formed on chlorination of o-xylene (o-dimethyl-benzene), m-xylene, and p-xylene?
-
When benzene is treated with D2SO4, deuterium slowly replaces all six hydrogens in the aromatic ring. Explain.
-
Which of the following alkyl halides would you expect to undergo Friedel-Crafts reaction without rearrangement? Explain. (a) CH3CH2Cl (b) CH3CH2CH (Cl) CH3 (c) CH3CH2CH2Cl (d) (CH3) CCH2Cl (e)...
-
Develop a 3-year financial projection for your proposed cancer tele-genetics program . State any and all assumptions you make in creating your estimates. Please consider the following in creating...
-
Lets assume that we have project L, which has a 40% chance of turning out good. The company Raises $7,000, 50% from bondholders demanding a 6% interest rate and 50% from stockholders, which becomes...
-
Please assist in the filling out the above and below table. I have provided all information that has been provided to me. Company Shares Outstanding Latest Share Price MVE Total Debt W d W e a b c =...

Study smarter with the SolutionInn App


