Question: Amine bases in nucleic acids can react with alkylating agents in typical SN2 reactions. Look at the following electrostatic potential maps, and tell which is
Amine bases in nucleic acids can react with alkylating agents in typical SN2 reactions. Look at the following electrostatic potential maps, and tell which is the better nucleophile, guanine or adenine. The reactive positions in each areindicated.
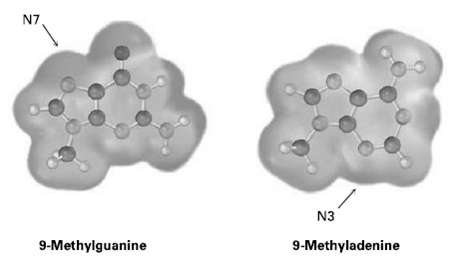
N7 N3 9-Methyladenine 9-Methylguanine
Step by Step Solution
3.42 Rating (171 Votes )
There are 3 Steps involved in it
According to the electrostatic potential map the ... View full answer

Get step-by-step solutions from verified subject matter experts
Document Format (1 attachment)
22-C-O-B-M (196).docx
120 KBs Word File


