An Assign E or Z configuration to each of the following alkenes: ( - C (a) H2
Question:
An Assign E or Z configuration to each of the following alkenes:
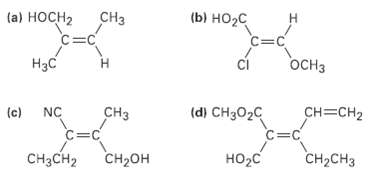
Transcribed Image Text:
(ы но-с CНз (a) НосH2 C=C C=C H3C ci OCH3 (c) (d) CH3O2C CH=CH2 NC CHз C=C CH3CH2 CH2он HO2C CH-CHз
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 71% (14 reviews)
c High HOCH CH3 High CC Low HC ...View the full answer

Answered By

Ehsan Mahmood
I’ve earned Masters Degree in Business Studies and specialized in Accounts & Finance. Couple with this, I have earned BS Sociology from renowned institute of Pakistan. Moreover, I have humongous teaching experience at Graduate and Post-graduate level to Business and humanities students along with more than 7 years of teaching experience to my foreign students Online. I’m also professional writer and write for numerous academic journals pertaining to educational institutes periodically.
4.90+
248+ Reviews
287+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Assign a systematic (IUPAC) name to each of the following compounds: (a) (b) (c) (d) (e) Br Br
-
Assign a priority order to each of the following sets of groups: a. -CH(CH3)2, -CH3, -H, -NH2 b. -OH, -Br, -CH3, -CH2OH c. -OCH3, -NH(CH3)2, -CH2NH2, -OH d. -CH2CH2CH3, -CH2CH3, -C(CH3)3, -CH(CH3)2
-
Determine the configuration of each of the following alkenes as Z or E as appropriate: (a) (b) (c) H3C CH2CH2F CH2CH2OH (CH3)3 C -C CH3CH2 CH3
-
Calculate the heat required to raise 50 kg of solid sodium carbonate (Na2CO3) from 10C to 50C at 1atm using (a) The true heat capacity of Na2CO3, which is 1.14 kJ / (kg C). (b) A heat capacity...
-
What are your best-developed leadership traits, motives, and characteristics? How do you know?
-
Can you foresee any problem with the write-once cache approach on bus-based multiprocessors? If so, suggest a solution.
-
What is the most accepted input power required for an ICP accelerometer?
-
The ledger of Rolling Hills Corporation contains the following accounts: Common Stock, Preferred Stock, Treasury Stock, Paid-in Capital in Excess of ParPreferred Stock, Paid-in Capital in Excess of...
-
Given the following annual returns for Stock ABC, what does a $3,000 investment grow to over the 5-year period? Year 1: 15% Year 2: 20% Year 3: 25% Year 4: 15% Year 5: 15%
-
Why is it reasonable to approximate H 11 and H 22 by the appropriate ionization energy of the corresponding neutral atom?
-
Rank the following sets of substituents in order of priority according to the Cahn-lngold?Prelog sequence rules: (a) -CH, -Br, -,-1 (b) -, -, -, -2 (c) -, -2CH3, -, - (d) -CH, -H2H, -H2H2, -H (e)...
-
Name the following cycloalkenes: (c) (b) (a) CH (f) (e) (d)
-
Suppose in our Universe there is only a monopole and antimonopole pair, situated at a distance \(\mathrm{d}\) of each other that is much smaller than the distance to any other atom in the Universe....
-
Except for the payment required, the activities of service wholesalers resemble the activities of what other type of wholesaler?
-
Who is considered a fiduciary under ERISA standards? Describe some of the standards and liabilities of fiduciaries.
-
Jamie has inherited 500 shares of an investment company. She calls her broker to redeem the shares and is informed that the kind of investment company she owns makes no provision for future purchases...
-
Affordable Beauty Supply produces inexpensive combs, brushes, and hair clips. As a way to minimize costs when sending out these small items, the company combines 10 to 12 different orders into one....
-
Calculate raw material cost per pound sold, for each source. 4. Calculate direct labor cost per pound sold for: a. Grading. b. Cutting. (consider yields carefully). 5. Calculate packing cost (labor...
-
When you pay an expense using cash, which account do you debit? The expense account or the cash account?
-
Define the term utility software and give two examples.
-
Determine the kinds of intermolecular forces that are present in each element or compound. a. PH 3 b. HBr c. CH 3 OH d. I 2
-
In the addition of HBr to 3,3-dimethyl-l-butene, the results observed are shown in Fig. P5.36. (a) Explain why the different conditions give different product distributions. (b) Write a detailed...
-
Give the structures of both the reactive intermediate and the product in each of the following reactions: (a) (b) CH,CH,CCH, + HBr- CH2 CH,CH CCHH rides
-
The bromine atom in the bromonium ion in Eq. 5.13 (p. lg3) has a positive formal charge. Why can't bromins- utrdergo a Lewis acid=base association reaction with a nucleophito?
-
Zach Ltd. produced the following data for the year 2015 - o Interest Income $4,650,000 o Capital Gain realized $50,000 o Dividend Income from company's 15% ownership in XYZ Ltd. $65,000 o Corporate...
-
A project requires an increase in net working capital of $150,000 at time 0 that will be recovered at the end of its 10 year life. If the opportunity cost of capital is 8%, what is the effect of NWC...
-
A firm is 40% financed by debt with a yield-to-maturity of 8.5%. The equity has a beta of 1.3, the market risk premium is 8.4% and the risk-free rate is 3.8%. What is the firm's WACC if the tax rate...

Study smarter with the SolutionInn App


