Question: As a general rule, equatorial alcohols are esterified more readily than axial alcohols. What product would you expect to obtain from reaction of the following
As a general rule, equatorial alcohols are esterified more readily than axial alcohols. What product would you expect to obtain from reaction of the following two compounds with 1 equivalent of aceticanhydride?
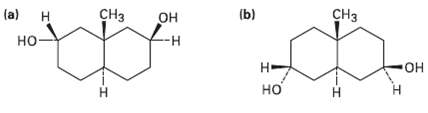
H (b) (a) CH - - -
Step by Step Solution
3.42 Rating (174 Votes )
There are 3 Steps involved in it
a H HO b H OH H CH3 H CH3 OH H H OH 1 eq... View full answer

Get step-by-step solutions from verified subject matter experts
Document Format (1 attachment)
22-C-O-B-M (167).docx
120 KBs Word File


