Question: As written, the following syntheses have flaws. What is wrong witheach? CH o (b) CI (a) .CH 1. Cl2, FeCl3 1. HNO3, H2SO4 2. CH3CI,
As written, the following syntheses have flaws. What is wrong witheach?
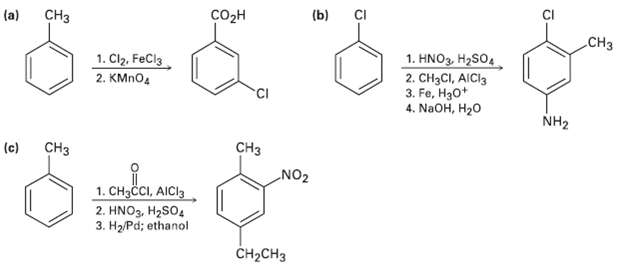
CH o (b) CI (a) .CH 1. Cl2, FeCl3 1. HNO3, H2SO4 2. CH3CI, AICI3 3. Fe, H30* 4. NaOH, H20 2. KMNO4 NH2 (c) CH3 CH NO2 1. CH3CI, AICI3 2. HNO3, H2SO4 3. H2/Pd; ethanol C-CH
Step by Step Solution
3.40 Rating (169 Votes )
There are 3 Steps involved in it
a Chlorination of toluene occurs at the ortho and Para positions To sy... View full answer

Get step-by-step solutions from verified subject matter experts
Document Format (1 attachment)
22-C-O-B (54).docx
120 KBs Word File


