At what approximate positions might the following compounds show IRabsorptions? (c) (b) CH CH (a) CH3CH2CH3 CHCCH2CH3CH2
Question:
At what approximate positions might the following compounds show IRabsorptions?
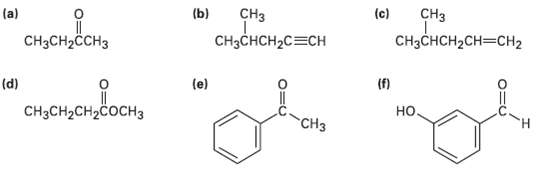
Transcribed Image Text:
(c) (b) CHз CHз (a) CH3CH2CH3 CHзCнCH2CH3CHН2 CHзснCH2C3CH (f) но. (d) (e) "онне CнзCH2CH2COCHз CHз
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 44% (9 reviews)
a b c d e Compound CH3CHCCH3 CH32CHCHCCH CH32CHCHCHCH CH3CHCHCOCH3 i CCH3 mol HO O Di...View the full answer

Answered By

Muhammad Ghyas Asif
It is my obligation to present efficient services to my clients by providing a work of quality, unique, competent and relevant. I hope you have confidence in me and assign me the order and i promise to follow all the instructions and keep time.
4.60+
109+ Reviews
203+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
At what approximate positions might the following compounds show IRabsorptions? c??H? " alt="At what approximate positions might the following compounds show IRabsorptions? 51524" class="fr-fic...
-
The following compounds show different rates of debromination. One reacts quite fast, and the other seems not to react at all. Explain this surprising difference in rates. Br KI, acetone Br (CH),C RS...
-
Which of the following compounds show only a single peak in their 1H NMR spectrum? a. CH3CH2OCH2CH3 b. c. CH,CH,CCI
-
Fill in the missing amounts for the following bank reconciliation: Practice Exercise3 Bank Reconciliation March 31,20 Bank Statement Balance $3,764.00 Add: Deposit in transit $4,031.00 Deduct:...
-
Why does Sutton's model apply so well to the consumer goods market? Does Sutton's model describe the structure of other markets?
-
The dual is constructed as shown in Fig. 8.118(a). The dual is redrawn as shown in Fig. 8.118(b). 0.25 0.5 2 4 1 9 A 9 A +3 V
-
Dry, compressed air at \(T_{m, i}=55^{\circ} \mathrm{C}, p=15 \mathrm{~atm}\), with a mass flow rate of \(\dot{m}=0.05 \mathrm{~kg} / \mathrm{s}\), enters a \(50-\mathrm{mm}-\) diameter,...
-
Financial information (in thousands of dollars) relating to three different companies follows. Instructions: 1. Compute the following ratios: (a) Return on sales (b) Asset turnover (c)...
-
Managing finances is essential for any business, big or small. It involves careful planning and allocation of resources to achieve goals. Budgeting is crucial as it helps firms manage their cash...
-
Prove that for a vacuum-dielectric interface at glancing incidence r ¥ -1, as in Fig. 4.49. Fig. 4.49. 1.0 0.5 -0.5 56.3 -1.0 30 90 0; (degrees) Amplitude coefficients
-
How would you use infrared spectroscopy to distinguish between the following pairs of constitutionalisomers? (a) CH3C=CCH3 and CHCH2C3CH () CHH CHCCH2HH2 and (c) H CH3CH2CHO and
-
Assume you are carrying out the dehydration of 1-methylcyclohexanol to yield 1-methykyclohexene. How could you use infrared spectroscopy to determine when the reaction is complete?
-
Imax Corporation is a large entertainment technology company, with headquarters in New York and Toronto, and theatres worldwide. Its share price, which was as high as Can.$ 13.89 on the Toronto Stock...
-
A bearish investor who wants to bet against a company can enter a short sale. Suppose you are bearish on XYZ stock, and its market price is $100 per share. You tell your broker to sell short 1,000...
-
Based on these exhibits, please make an Excel template for analysis of the Redhook Ale Brewery loan application . Specifically, what do you think of David Mickelson's projections? Hint: consider the...
-
In your response, include an example of a business that has suffered from poorly forecasting the demand of its products. Evaluate how or why the business made such a mistake. Why is knowing (or...
-
1. It is found that the equilibrium concentration of the undissociated form (HA) of a weak monoprotic acid, in an aqueous solution of pH-3, is 9.398x104 mol/L. It is also known that an equal amount...
-
Provide the missing information. (Always use cell references and formulas where appropriate to receive full credit. If you copy/paste from the Instructions tab you will be marked wrong.) Item Case 1...
-
You are in charge of designing a new hazardous waste landfill for Kiev. You are trying to decide whether it is worth spending R 1,000,000 to install a special liner that reduces the probability of a...
-
In the simple quantity theory of money, what will lead to an increase in aggregate demand? In monetarism, what will lead to an increase in aggregate demand?
-
An isotonic solution contains 0.90% NaCl mass to volume. Calculate the percent mass to volume for isotonic solutions containing each solute at 25 C. Assume a vant Hoff factor of 1.9 for all ionic...
-
Draw the organic products you would expect to isolate from the following reactions (after hydrolysis). (a) (b) (c) (d) (e) (f) (g) (h) (i) (j) (k) (m) (n) (o) (CH2 = CH)2 CuLi + CH3CH2CH= CHCH2Br...
-
Show how you would use Grignard syntheses to prepare the following alcohols from the indicated starting materials and any other necessary reagents. (a) octan-3-ol from hexanal, CH3 (CH2)4CHO (b)...
-
Give a systematic (IUPAC) name for each diol. (a) CH3CH(OH)(CH2)4 CH(OH)C(CH3)3 (b) HO--(CH2)8-OH (C) (d) (e) HO HO
-
1. Explain what the "Deinstitutionalization Mandate" is. This should be done in at least two paragraphs. 2. In another two paragraphs, discuss how status offenses are connected to the...
-
You are the owner of a very small business that sells gourmet coffee. You sell only one product, a 12-ounce bag of whole-bean French roast coffee. You sell each bag of coffee for $14 each, but due to...
-
Find the instantaneous rate of change of the function f(t)=3+5t when t=-3.

Study smarter with the SolutionInn App


