Benzyl chloride can be converted into benzaldehyde by treatment with nitro methane and base. The reaction involves
Question:
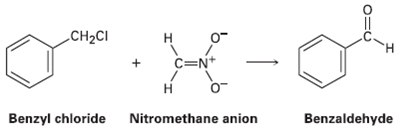
Benzyl chloride can be converted into benzaldehyde by treatment with nitro methane and base. The reaction involves initial conversion of nitro methane into its anion, followed by SN2 reaction of the anion with benzyl chloride and subsequent E2 reaction. Write the mechanism in detail, using curved arrows to indicate the electron flow in each step.
Transcribed Image Text:
CH2CI н C=N+ Н Benzyl chloride Nitromethane anion Benzaldehyde
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 77% (9 reviews)
09100 CH 0 ...View the full answer

Answered By

Carly Cimino
As a tutor, my focus is to help communicate and break down difficult concepts in a way that allows students greater accessibility and comprehension to their course material. I love helping others develop a sense of personal confidence and curiosity, and I'm looking forward to the chance to interact and work with you professionally and better your academic grades.
4.30+
12+ Reviews
21+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Using curved arrows to indicate the election flow in each step, show how the base-catalyzed reverse aldol reaction of 4-hydroxy-4-methly-2-pentanone takes place to yield 2 equivalents of acetone.
-
Benzyl bromide is converted into benzaldehyde by heating in dimethyl sulfoxide. Propose a structure for the intermediate, and show the mechanisms of the two steps in thereaction. CH2BR E2 reaction...
-
Show how 4-methylcyclohexyl chloride can be converted to 4- methylcyclohexanecarboxylic acid.
-
IFRS requires the following: (a) Ending inventory is written up and down to net realizable value each reporting period. (b) Ending inventory is written down to net realizable value but cannot be...
-
The model below depicts industry analysis as a funnel that focuses on remote-factor analysis to better understand the impact of factors in the operating environment. Do you find this model...
-
Using the annual report of any entity of your choice, discuss whether the information provided therein is of use to users.
-
This is a continuation of comprehensive question. 1. Since the Robinson Company is short of cash, the deposit of receipts is also expedited. The receptionist turns over all mail receipts and related...
-
Karane Enterprises, a calendar-year manufacturer based in College Station, Texas, began business in 2017. In the process of setting up the business, Karane has acquired various types of assets. Below...
-
The system call sbrk (0) returns program crash, if it does? 1 2 3 4 6 7 8 } T-W21-DPe current value of brk. When will the f #include int main() { char *c; c = sbrk (0) - 1; *C= k'; c = sbrk (0) + 1;...
-
The following information pertains to Torasic Companys budgeted income statement for the month of June 2011: Sales (1,200 units at $250) $300,000 Variable cost 150,000 Contribution margin $150,000...
-
Rank the following substituted phenols in order of increasing acidity, and explain your answer: CH
-
Reduction of 2-butanone with NaBH4 yields 2-butanol. Is the product chiral is it optically active? Explain.
-
You and a friend are in the same accounting class. During the first test, you observe that your friend cheated by copying one of her answers from another student (who was unaware of the copying)....
-
On December 3 1 , 2 0 2 4 , the end of the fiscal year, California Microtech Corporation completed the sale of its semiconductor business for $ 1 8 million. The semiconductor business segment...
-
Kyler ( ordinary rate of 3 5 % in this question; regular capital gains rate of 1 5 % ) purchased Wong stock for $ 4 0 0 in February of 2 0 2 2 . In November of 2 0 2 2 , the stock had appreciated to...
-
Actuarial Science An insurer is reviewing claims for a certain line of insurance from Accident year 2 0 2 3 . The earned premiums in 2 0 2 3 were $ 7 . 7 million. The base premium in 2 0 2 3 was $ 1...
-
he Data Group Inc. had the following balances in its equity accounts at December 3 1 , 2 0 2 2 : Common shares, unlimited shares authorized; 2 1 0 , 0 0 0 shares issued and outstanding $ 3 , 9 9 0 ,...
-
Ida Sidha Karya Company is a family - owned company located in the village of Gianyar on the island of Bali in Indonesia. The company produces a handcrafted Balinese musical instrument called a...
-
Which of the following statements correctly describes the reporting of cash? a. Cash cannot be combined with cash equivalents. b. Restricted cash need not be disclosed. c. Cash is listed last in the...
-
Refer to the information from Exercise 22-19. Use the information to determine the (1) Weighted average contribution margin , (2) Break-even point in units, and (3) Number of units of each product...
-
The enthalpy of formation of NaI(s) is -288 kJ mol -1 . Use this value, together with other data in the text, to calculate the lattice energy of NaI(s).
-
List the following compounds in order of decreasing reactivity toward CH3O- in an SN2 reaction carried out in CH3OH: CH3F, CH3Cl, CH3Br, CH3I, CH3OSO2CF3, 14CH3OH.
-
Starting with (S)-2-bromobutane, outline syntheses of each of the following compounds: (a) (b) (c) (d) (R)-CH3CHCH2CH3 OCH2CH3 (R)-CH3CHCH2CH3 CCH3 (R)-CH3CHCH2CH3 SH (R)-CH3CHCH2CH3 SCH3
-
Which alkyl halide would you expect to react more rapidly by an SN2 mechanism? Explain your answer. (a) (b) (c) (d) (e) Br r or or CI
-
Determine three planes) interection point. Th = 5x+4y+52-18=0 712=4x-54+9=0 13=-5x+10y-12=0
-
A particle initially has a speed of 0.46c. (Enter your answers in terms of c. Round your answers to at least three decimal places.) (a) At what speed does its momentum increase by 1%? .465 x c (b) At...
-
5) Megan and four of her friends plan to go for a long drive. She buys 12 bottles of frappe and a few bottles of orange juice from a supermarket. The total number of bottles they have are four times...

Study smarter with the SolutionInn App


