Benzyl chloride can be converted into benzaldehyde by treatment with nitro methane and base. The reaction involves
Question:
Benzyl chloride can be converted into benzaldehyde by treatment with nitro methane and base. The reaction involves initial conversion of nitro methane into its anion, followed by SN2 reaction of the anion with benzyl chloride and subsequent E2 reaction. Write the mechanism in detail, using curved arrows to indicate the electron flow in each step.
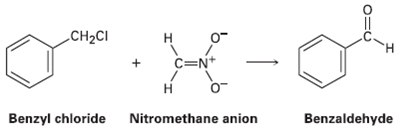
Transcribed Image Text:
CH2CI н C=N+ Н Benzyl chloride Nitromethane anion Benzaldehyde
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 77% (9 reviews)
09100 CH 0 ...View the full answer

Answered By

Carly Cimino
As a tutor, my focus is to help communicate and break down difficult concepts in a way that allows students greater accessibility and comprehension to their course material. I love helping others develop a sense of personal confidence and curiosity, and I'm looking forward to the chance to interact and work with you professionally and better your academic grades.
4.30+
12+ Reviews
21+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Using curved arrows to indicate the election flow in each step, show how the base-catalyzed reverse aldol reaction of 4-hydroxy-4-methly-2-pentanone takes place to yield 2 equivalents of acetone.
-
Benzyl bromide is converted into benzaldehyde by heating in dimethyl sulfoxide. Propose a structure for the intermediate, and show the mechanisms of the two steps in thereaction. CH2BR E2 reaction...
-
Show how 4-methylcyclohexyl chloride can be converted to 4- methylcyclohexanecarboxylic acid.
-
IFRS requires the following: (a) Ending inventory is written up and down to net realizable value each reporting period. (b) Ending inventory is written down to net realizable value but cannot be...
-
The model below depicts industry analysis as a funnel that focuses on remote-factor analysis to better understand the impact of factors in the operating environment. Do you find this model...
-
The global economy has seen a fall in the number of landlines and rapid growth in the number of smartphones. In the United States, 41 percent of households have no landline and 90 percent have a...
-
If among \(n\) objects \(k\) are alike and the others are all distinct, the number of permutations of these \(n\) objects taken all together is \(n ! / k !\). (a) How many permutations are there of...
-
In the past, patrons of a cinema complex have spent an average of $5.00 for popcorn and other snacks, with a standard deviation of $1.80. The amounts of these expenditures have been normally...
-
1. Comment each line of this HTML/JavaScript code to describe what it is doing. 2. Sketch a picture of the webpage before the button is clicked. 3. Sketch a picture of the web page after the button...
-
The disclosure rules for business combinations complicate financial analysis. Trend analysis becomes difficult because comparative financial statements are not retroactively adjusted to include data...
-
Rank the following substituted phenols in order of increasing acidity, and explain your answer: CH
-
Reduction of 2-butanone with NaBH4 yields 2-butanol. Is the product chiral is it optically active? Explain.
-
Calculate the mean deviation for Doty Hall, and compare it to (as calculated in Exercise 7). What do you think would happen to the difference of these two measures if you were to add an extreme...
-
explain why the type of becerage sold is an example of a catwgorical variable.
-
The premium on a $75,000 fire insurance policy from January 1, 2015 to January 1, 2018 10. is $900. On December 30, 2015, there was a fire loss of $50,000, what was subsequently paid by the insurance...
-
You are in charge of forecasting revenue for the government. Assume the government is going to increase the tax rate. What type of taxpayer is likely to respond to this tax rate increase by working...
-
Explain the term business risk, and suggest the measures that should be adopted by managers in mitigating against this risk.
-
How can evaluative frameworks and outcome metrics be utilized to assess the impact of empowerment initiatives, including measures of individual well-being, social capital, collective efficacy, and...
-
Do you think the ability of a convicted defendant to secure DNA testing rises to an issue of due process of law? Does Congress have a role to play in providing a standardized process throughout the...
-
Refer to the information from Exercise 22-19. Use the information to determine the (1) Weighted average contribution margin , (2) Break-even point in units, and (3) Number of units of each product...
-
The enthalpy of formation of NaI(s) is -288 kJ mol -1 . Use this value, together with other data in the text, to calculate the lattice energy of NaI(s).
-
List the following compounds in order of decreasing reactivity toward CH3O- in an SN2 reaction carried out in CH3OH: CH3F, CH3Cl, CH3Br, CH3I, CH3OSO2CF3, 14CH3OH.
-
Starting with (S)-2-bromobutane, outline syntheses of each of the following compounds: (a) (b) (c) (d) (R)-CH3CHCH2CH3 OCH2CH3 (R)-CH3CHCH2CH3 CCH3 (R)-CH3CHCH2CH3 SH (R)-CH3CHCH2CH3 SCH3
-
Which alkyl halide would you expect to react more rapidly by an SN2 mechanism? Explain your answer. (a) (b) (c) (d) (e) Br r or or CI
-
Sweeten Company had no jobs in progress at the beginning of the year and no beginning inventories. It started, completed, and sold only two jobs during the year-Job P and Job Q. The company uses a...
-
1. Review the following tracings, identify the type of BBB, and then explain how you determined your answers 2. Review the following tracing. Do you suspect left ventricular hypertrophy? Explain how...
-
What are signs that its time to cut corners to get the product launched, and what would you cut?

Study smarter with the SolutionInn App


