Question: Outline a synthesis of the quaternary ammonium salt (CH 3 ) 3 Br from each of the following combinations of starting materials. Benzylamine and
Outline a synthesis of the quaternary ammonium salt (CH3)3 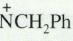 Br– from each of the following combinations of starting materials.
Br– from each of the following combinations of starting materials.
Benzylamine and any other reagents
NCH,Ph
Step by Step Solution
3.43 Rating (166 Votes )
There are 3 Steps involved in it
Exhaustively methylate be... View full answer

Get step-by-step solutions from verified subject matter experts
Document Format (1 attachment)
902-O-EE (288).docx
120 KBs Word File


