Question: Calculate No (E), the density of occupied states, for copper at T = 1000 K for an energy E of(a) 4.00eV(b) 6.75 eY(c) 7.00 eV(d)
Calculate No (E), the density of occupied states, for copper at T = 1000 K for an energy E of(a) 4.00eV(b) 6.75 eY(c) 7.00 eV(d) 7.25 eV, and(e) 9.00 eV. Compare your results with the graph of Figure. The Fermi energy for copper is7.00eV.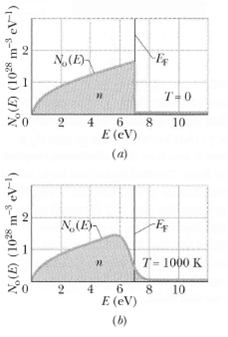
N,(E) T=0 10 0. 8. E (cV) 2. (a) N,(E) T = 1000 K 6 8 E (eV) 2. 10 (b) N,(E) (102* m eV-l) (1028 m-3 eV-l) 4,
Step by Step Solution
★★★★★
3.27 Rating (159 Votes )
There are 3 Steps involved in it
1 Expert Approved Answer
Step: 1 Unlock

We use NE NEPE CE2 eEEp 1 where see Problem 413 827m2 827910910 kg 2 h 6626x1034 Js3 681x... View full answer

Question Has Been Solved by an Expert!
Get step-by-step solutions from verified subject matter experts
Step: 2 Unlock
Step: 3 Unlock
Document Format (1 attachment)
18-P-M-P-C-E (15).docx
120 KBs Word File


