Calculate the pressure exerted by 0.5000 mole of N 2 in a 1.0000-L container at 25.0 o
Question:
Calculate the pressure exerted by 0.5000 mole of N2 in a 1.0000-L container at 25.0oC. (See Table)
a. Use the ideal gas law.
b. Use the van der Waals equation.
c. Compare the results from parts a and b.
Table
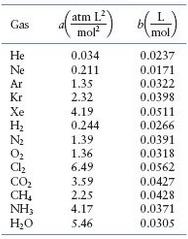
Transcribed Image Text:
\ 7 1 200 1 6 1 8 2 7815 2133 (000000000000 691622 23354433 LF- 11 5 2 9 4 9699576 345214 m | 0 2 3 3 1 2 3 0012401163245
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 37% (8 reviews)
a b c PV nRT P n...View the full answer

Answered By

Bhartendu Goyal
Professional, Experienced, and Expert tutor who will provide speedy and to-the-point solutions. I have been teaching students for 5 years now in different subjects and it's truly been one of the most rewarding experiences of my life. I have also done one-to-one tutoring with 100+ students and help them achieve great subject knowledge. I have expertise in computer subjects like C++, C, Java, and Python programming and other computer Science related fields. Many of my student's parents message me that your lessons improved their children's grades and this is the best only thing you want as a tea...
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Physical Chemistry questions
-
Calculate the pressure exerted by 1.0 mol H2S behaving as (a) A perfect gas, (b) A van der Waals gas when it is confined under the following conditions: (i) At 273.15 K in 22.414 dm3, (ii) At 500 Kin...
-
Van der Waals Equation and Critical Points (a) In p V- diagrams the slope p/V along an isotherm is never positive. Explain why. (b) Regions where p/V = 0 represent equilibrium between two phases;...
-
By what magnitude would the pressure exerted by water on the walls of the vessel have increased if the intermolecular attraction forces had vanished?
-
Assume that your team has been in contract with the headquarters of a company that owns several restaurants in different states in the US. Your team is to provide software that manages these...
-
Allies Apples, Inc. purchases apples in bulk and sells two products, boxes of apples and jugs of cider. Allies has capacity limitations of three kinds: warehouse space, crating facilities, and...
-
Suppose you have the opportunity to make an investment in a real estate venture that expects to pay investors $750 at the end of each month for the next eight years. You believe that a reasonable...
-
Let \(q(t)\) be the survival probability and let \(q^{-1}\) be its inverse function. Also, let \(U\) be a uniform random variable on \([0,1]\). For each realization \(u\), let \(\tau\) be chosen such...
-
Company A has three debt issues of $3,000 each. The interest rate on issue A is 4 percent, on B the rate is 6 percent, and on C the rate is 8 percent. Issue B is subordinate to A, and issue C is...
-
what ways does the symbiotic relationship between mentor and mentee transcend conventional paradigms, fostering a profound interchange of knowledge, wisdom, and experiential insight to sculpt...
-
Write the Fourier series of the following function on the given intervals. i. f (x) = 1 - |x|, -2 < x < 2 {-4, for -ns x <0 4, for 0 sxsn ii. f(x) = iii. f (x) = sin(2x), - n < x s n iv. f (x) = x2 -...
-
It took 4.5 minutes for 1.0 L of helium to effuse through a porous barrier. How long will it take for 1.0 L of Cl 2 gas to effuse under identical conditions?
-
Calculate the pressure exerted by 0.5000 mole of N 2 in a 10.000- L container at 25.0 o C. (See Table) a. Use the ideal gas law. b. Use the van der Waals equation. c. Compare the results from parts a...
-
The probability that a person does his or her own return is 0.85. The probability that a person receives a refund is 0.76. The probability that a person does his or her own return and receives a...
-
Weatherford Industries Inc. has the following ratios: A 0 */S 0 = 1.6; L 0 */S 0 = 0.4; profit margin = 0.10; and payout ratio = 0.45, or 45%. Sales last year were $100 million. Assuming that these...
-
Carlsbad Corporations sales are expected to increase from $5 million in 2021 to $6 million in 2022, or by 20%. Its assets totaled $3 million at the end of 2021. Carlsbad is at full capacity, so its...
-
Kahn Inc. has a target capital structure of 60% common equity and 40% debt to fund its $10 billion in operating assets. Furthermore, Kahn Inc. has a WACC of 13%, a before-tax cost of debt of 10%, and...
-
The Paulson Companys year-end balance sheet is shown here. Its cost of common equity is 14%, its before-tax cost of debt is 10%, and its marginal tax rate is 25%. Assume that the firms long-term debt...
-
Hook Industriess capital structure consists solely of debt and common equity. It can issue debt at r d = 11%, and its common stock currently pays a $2.00 dividend per share (D 0 = $2.00). The stocks...
-
Why are cohesive groups important to us as individuals? What are some reasons managers should care about the presence of groups and group cohesiveness?
-
Distinguish among total-moisture content, free-moisture content, equilibrium-moisture content, unbound moisture, and bound moisture.
-
A certain oxide of titanium is 28.31% oxygen by mass and contains a mixture of Ti2+ and Ti3+ ions. Determine the formula of the compound and the relative numbers of Ti2+ and Ti3+ ions.
-
Spinel is a mineral that contains 37.9% aluminum, 17.1% magnesium, and 45.0% oxygen, by mass, and has a density of 3.57 g/cm3. The edge of the cubic unit cell measures 809 pm. How many of each type...
-
A metallic solid with atoms in a face-centered cubic unit cell with an edge length of 392 pm has a density of 21.45 g/cm3. Calculate the atomic mass and the atomic radius of the metal. Identify the...
-
What theorists are most likely to argue the city's form and growth come from decisions made by people and organizations that control wealth and other key resources?
-
Explain the difference between glucogenic and ketogenic amino acids
-
Describe what is an arrangement where workers who don't join a union must make payments equal to union dues and fees to get union representation services?

Study smarter with the SolutionInn App


