Cis-4-Octene reacts with a peroxyacid to yield 4, 5-epoxyoctane. Is the product chiral? How many chirality centers
Question:
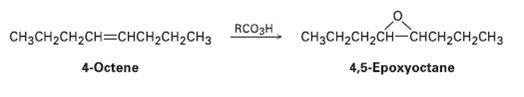
Cis-4-Octene reacts with a peroxyacid to yield 4, 5-epoxyoctane. Is the product chiral? How many chirality centers does it have? How would you describe it stereo chemically?
Transcribed Image Text:
снснс CH3CH2CH2CH=CHCH2CH2CH3 CH3CH2CH2CH-CHCH2CH2CH3 RCO3H 4-Octene 4,5-Ерохуосtane
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 44% (18 reviews)
R CHCHCHCHCHCH3 H CHCHCH O R CHCHCH3 Peroxycarboxylic acids ...View the full answer

Answered By

Muhammad Umair
I have done job as Embedded System Engineer for just four months but after it i have decided to open my own lab and to work on projects that i can launch my own product in market. I work on different softwares like Proteus, Mikroc to program Embedded Systems. My basic work is on Embedded Systems. I have skills in Autocad, Proteus, C++, C programming and i love to share these skills to other to enhance my knowledge too.
3.50+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
How many chirality centers are contained in (a) The aldotetrose (b) The ketopentose just given? (c) How many stereoisomers would you expect from each general structure?
-
How many chirality centers are present in estradiol how many stereo isomers does estradiol have?
-
How many chiral carbon atoms does the following structure have? ,
-
Explain, in your own words, what is meant by source inspection.
-
In one of the photos at the beginning of this chapter, Mary Beth uses a torque feeler that consists of a meter stick held at the 0-cm end with a weight dangling from various positions along the...
-
A beaker of water at room temperature is placed in an enclosure, and the air pressure in the enclosure is slowly reduced. When the air pressure is reduced sufficiently, the water begins to boil. The...
-
Suppose that your company is trying to decide whether it should buy special equipment to prepare high-quality publications itself or lease the equipment from another company. Suppose that leasing the...
-
Warner Co. entered into the following transactions involving short-term liabilities in 2016 and 2017. 2016 Apr. 22 Purchased $5,000 of merchandise on credit from Fox-Pro, terms n30. Warner uses the...
-
On July 1, year 1, ABC. Ltd. purchased a piece of equipment for $ 500000 plus installation costs of $ 60000 that were necessary to make the equipment operational. ABC. Ltd. has a December 31, fiscal...
-
Franz Manteca is the president of Gillespie Storage, a wholly owned subsidiary of Vanguard Industries. His annual salary is $600,000. In addition, he receives a $200,000 bonus if Gillespie Storages...
-
Hydroxylation of trans-2-butene with OsO4 also yields 2, 3-butanediol. What stereochemistry do you expect for the product?
-
Answer Problem 9.61 for the epoxidation of trans-4-octene. CH3CH2CH2CH=CHCH2CH2CH3 CH3CH2CH2CH-CHCH2CH2CH3 RCO3H 4-Octene 4,5-tane
-
A 2-MVA transformer is open for procurement. Vendor A offers 96.5% efficiency at $160,000 price, and vendor B offers 97.8% efficiency at $180,000 price. Determine the present worth of buying the...
-
Think back to a time you experienced a communication breakdown in a personal or social setting (something you're comfortable discussing with the class in a public forum). 1. Did you figure out why...
-
Imagine you are visiting your aunt, who is a patient in a hospital in a nearby city. While you are sitting at her bedside, you hear a lot of noise at the nurses' station, as if they are having a...
-
Using Houseplan #5 on page 4 of the Measurement supplement(below), determine the cost of pouring the 9 inch thick concreteslab for this home, assuming that the porch will also be on thefoundation....
-
Recall from lecture that Flip-Flap Railway is an old roller coaster that was built in a circle. It has a diameter of 25 ft and riders entered the ride at a speed of 45 mph. At the top of the loop,...
-
Small Fry Design, founded in 1997, is a toy and accessories company that designs and imports products for children. The company's line of merchandise includes teddy bears, musical toys, rattles and...
-
What is the Club Managers Association of America? LO.1
-
Suppose the spot and six-month forward rates on the Norwegian krone are Kr 5.78 and Kr 5.86, respectively. The annual risk-free rate in the United States is 3.8 percent, and the annual risk-free rate...
-
Treatment of gold metal with BrF 3 and KF produces Br 2 and KAuF 4 , a salt of gold. Identify the oxidizing agent and the reducing agent in this reaction. Find the mass of the gold salt that forms...
-
Using bases (B:) and acids (+BH) as needed, provide a curved-arrow mechanism for the conversion of the c-amino acid serine into formaldehyde and glycine (Eq. 25.53, p. 1242). Eq. 25.53 formaldehyde...
-
Draw the structure of GDP..
-
Draw in detail the structure of a section of RNA four residues long that, from the 5'-end, has the following sequence of bases: A, U, C, G. Label the 3' and 5' ends.
-
explain in excel please For a particular product the price per unit is $6. Calculate Revenue if sales in current period is 200 units. Conduct a data analysis, on revenue by changing the number of...
-
Hall Company sells merchandise with a one-year warranty. In the current year, sales consist of 35,000 units. It is estimated that warranty repairs will average $10 per unit sold and 30% of the...
-
Q 4- Crane Corporation, an amusement park, is considering a capital investment in a new exhibit. The exhibit would cost $ 167,270 and have an estimated useful life of 7 years. It can be sold for $...

Study smarter with the SolutionInn App


