Classify each of the labeled bonds in the following structure in terms of the bond type (
Question:
Classify each of the labeled bonds in the following structure in terms of the bond type (σ or π) and the component orbitals that overlap to form the bond. (For example, the carbon--carbon bond in ethane is an sp3-sp3 a bond.)
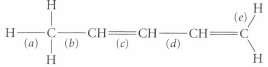
Transcribed Image Text:
(el ta) (b) lc)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 87% (8 reviews)
a An sp 3 C1sH s bond b An sp ...View the full answer

Answered By

Jason J Pulikkottil
I am currently pursuing a Master's degree in Computer Applications.I have completed a professional diploma in IT infrastructure management.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Classify each of the following statements as true or false: (a) For molecular solids the melting point generally increases as the strengths of the covalent bonds increase. (b) For molecular solids...
-
Classify each of the following as a primary, secondary, or tertiary alcohol. a. b. c. d. CH3 CH3 CH3CCH2CHOH CH CH,OH CH,CH2CCH CH3 CH2CH3 CH3CH2COH CH2CH3
-
Classify each of the following statements as true or false: (a) Although both molecular solids and covalent-network solids have covalent bonds, the melting points of molecular solids are much lower...
-
The manufactured wood beam carries a uniformly distributed load of in- tensity wo. Determine the largest safe value of wo if the maximum shear stress in the wood is limited to 300 psi. 1.0 ft Wo 4 ft...
-
Consider an argument you have recently had with a friend, family member, manager, co-worker, or someone else. Identify the topic of the argument and present that argument in premise-conclusion form,...
-
A study by Becker Associates, a San Diego travel consultant, found that 30% of the traveling public said that their flight selections are influenced by perceptions of airline safety. Thirty-nine...
-
It is claimed that problem definition and decision-making are intertwined. Explain.
-
Select any three countries that might have some logical basis for establishing a multinational market organization and illustrate their compatibility as a regional trade group. Identify the various...
-
Ex1: Suppose you have these related classes: class Pet { private String name; public String getName() { return name; } public void setName(String petName) { name = petName; } public String speak() {...
-
At 45C, 1,000 kg/h of a mixture of 0.80 mass fraction docosane and 0.20 mass fraction diphenylhexane is extracted with pure furfural to remove some diphenylhexane from the feed. Phaseequilibrium data...
-
Classify the compounds within each of the following pairs as either identical molecules (I), constitutional isomers (C), stereoisomers (S), or none of the above (N). (a) cyclopentane and cyclopentene...
-
Give the structure of: 2E,7Z)-5-[(4- 1-propenyl]-2,7 -non adiene Be sure to read Study Guide Link 4.2 if you have difficulty with this problem.
-
The name Prepaid Expense suggests that this account is an expense and belongs on an income statement. Instead the account appears on the balance sheet as an asset. Explain why this is appropriate and...
-
Discuss which strategy each of the companies profiled in the videos below used. Why Starbucks Failed In Australia? Why 7-Eleven Failed In Indonesia? Why McDonald's Flopped In Vietnam?
-
Find what you believe is an effective video/oral or oral marketing ad on the web. Provide the link as a part of this response. Utilizing concepts of marketing as context, provide your thoughts as to...
-
Pick a US-based, publicly traded insurance company. You are to produce a report about the company you chose that includes the following: Company News A Chart of the Company's Stock Performance...
-
describe the incident of Texas infrastructure collapse during the freeze, and then discuss the professional response to it. a. Critically analyze what went right and what went wrong with the...
-
Describe the issues surrounding the monitoring of venue customer service, in particular, the application of venue standards by various members of a management team. Your answer should include...
-
International travel is usually more expensive than domestic travel. A recent survey found that the average per-person cost of a 12-day international vacation is $1,755. This includes transportation,...
-
Independent random samples of sizes n1 = 30 and n2 = 50 are taken from two normal populations having the means 1 = 78 and 2 = 75 and the variances 21 = 150 and 22 = 200. Use the results of Exercise...
-
Which of the following orbitals is (are) not permitted by the quantum theory of the hydrogen atom? Explain. 2s 6s 5d 2d 3p
-
Give the electronic configuration of (a) The chlorine atom; (b) The silicon atom (Si); (c) The argon atom; (d) The magnesium atom. Indicate the valence electrons and the valence orbitals of Si.
-
Predict the approximate bond angles in each of the following molecules, and explain your reasoning. (a) :CH (b) BeH (c) *CH3 (d):CLSi (e) -0-0 (f) HC=C=CH allene HC-N OF ozone :O: (Give H-C-C and...
-
The quality-control department of Starr Communications, the manufacturer of video-game DVDs, has determined from records that 1.6% of the DVDS sold have video defects, 0.9% have audio defects, and...
-
What is the half-life in hours of procainamide in a patient whose total clearance is estimated to be 20 L/h and the volume of distribution is 120 L assuming one compartment, open model, first- order...
-
1. The technologies, such as ChatGPT, has been launched before introducing even a baseline of safety measures. This goes back to trade-offs that companies make, and people just watch the impact from...

Study smarter with the SolutionInn App


