Complete the electron-dot structure of caffeine, showing all lone-pair electrons, and identify the hybridization of the indicatedatoms.
Question:
Complete the electron-dot structure of caffeine, showing all lone-pair electrons, and identify the hybridization of the indicatedatoms.
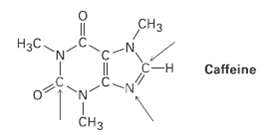
Transcribed Image Text:
CHз H3C. -- Caffeine 0= Cнз
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 70% (17 reviews)
HC 0 0 CH3 CH3 ...View the full answer

Answered By

Deepak singh
I have completed my graduation from computer science engineering . I have good knowledge about programming language and love to solve many codes of any language. I have been an expert to other platforms too and have good experience of giving solution to the students of subject .
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Identify all nonbonding lone pairs of electron in the following molecules, and tell what geometry you expect for each pf the indicated atoms. (a) The oxygen atom in the dimethyl ether, CH3 ? O ? CH3...
-
The file Caffeine contains the caffeine content (in milli-grams per ounce) for a sample of 26 energy drinks: a. Construct an ordered array. b. Construct a stem- and- leaf display. c. Does the ordered...
-
Identify the expected hybridization state and geometry for the central atom in each of the following compounds: a. b. c. d. e. H. I-z: I-
-
Suppose that Marthas income rises to $42,000 per year, and that she increases her consumption of health care visits by fi ve visits. Using the graphs for Exercise 1, draw the new equilibrium. What is...
-
Dedriea contributes to her wholly owned corporation some tangible personal property that she had used in her sole proprietorship business and depreciated. She had acquired the property for $566,000...
-
Blood plasma proteins of patients with malignant breast tumors differ from proteins of healthy people in their solubility in the presence of various polymers. When the polymers dextran and...
-
An airplane is flying at a flight (or local) Mach number of 0.70 at \(10,000 \mathrm{~m}\) in the Standard Atmosphere. Find the ground speed (a) if the air is not moving relative to the ground (b) if...
-
Hugo Reyes Company had the following account balances at year-end: Cost of Goods Sold $60,000; Inventory $15,000; Operating Expenses $29,000; Sales Revenue $115,000; Sales Discounts $1,200; and Sales...
-
Answer the below 2 questions separately on how to set, plan, and obtain goals. 1. Using the SMART framework, create 3 Goals: 1 personal, 1 professional, 1 academic. Make sure you use the SMART...
-
Ladysmith Furnishings extends generous payment terms to its customers. As a result, it has accumulated a substantial portfolio of accounts receivable. To improve cash flow, the company decided to...
-
Allene (see problem 1.46) is related structurally to carbon dioxide, CO2. Draw a picture showing the orbitals involved in the and bonds CO2, and identify the likely hybridization of carbon.
-
Almost all stable organic species have tetravalent carbon atoms, but species with trivalent carbon atoms also exist. Carbocations are one such class of compounds. (a) How many valence electrons do...
-
Fill in the blanks to indicate how investments are reported under IFRS. a. Financial instruments are measured at ______________. b. Gains or losses on equity investments with no significant influence...
-
Concepts of Positive Psychology . Find a website that reflects important positive psychology concepts and that would contribute to a person's overall well-being. Post a link to this website. Provide...
-
Draw an indifference map for each: Part A: Suppose that a labor union cares only about a wage payment to its member. Part B: Now the labor union cares only about the employment level of its members....
-
research and explain the history, common focus/topics, and information on positive psychology as if to explain to a person who has never heard of it. You will need a minimum of 5 references for this...
-
What is the role of empowerment in Talent Management in the era of Talent Empowerment? What do you think are the best practices that companies can implement to empower their talented employees...
-
A paralegal has been served with a court order that requires you to attend at court to produce certain documents and information regarding one of your clients. Do you need your client's consent?...
-
Which of the following is consistent with the expectation that exploiting a structural inefficiency is repeatable? A. The inefficiency is a unique event that occurs infrequently. B. The level of...
-
Discuss the information available from the following techniques in the analysis of inorganic pigments used in antique oil paintings: (i) Powder X-ray diffraction, (ii) Infrared and Raman...
-
The vant Hoff corollary to the third law of thermodynamics is that whenever two solid forms of a substance are known, the one with the greater specific heat will be the more stable one at higher...
-
Draw structure for the neutral molecule represented by the following model. Explain whether the octet rule is satisfied at each atom of the compound. Draw all of the important resonance structures...
-
Indicate whether each of these species can act as an acid, a base, or both: H +1 a) H-N-H 1 H H e) H-C-O-H H b) H-O-H H: H TI f) H-C-C-C-H H H c) H-C-H H H d):Ci : g) H-O-C-:
-
Show the conjugate acids of these species: a) CH-O-H b) H-O: C) CH,NH,
-
12 Stiler XYZ currently has an enterprise value of $600 million, 20 million shares outstanding, $200 million in excess cash and no debt. Assuming XYZ uses its excess cash to repurchase shares, and...
-
Explain a summary of the news story in the link below. Your summary should include at least 3 of the marketing concepts learned to date in this course. Be sure to include an accurate, succinct...
-
Salmon ASA has just issued a callable seven-year, 8% coupon bond with coupon payable annually. The bond can be called at par in two years or anytime thereafter on a coupon payment date. It has a...

Study smarter with the SolutionInn App


