Question: Convert the following shorthand representations to structures showing all of the atoms, bonds, and unshared electron pairs: d) COOH COH b) OH de c) f)
Convert the following shorthand representations to structures showing all of the atoms, bonds, and unshared electron pairs:
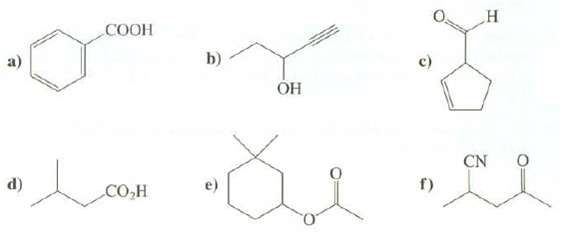
d) COOH COH b) OH de c) f) H CN Ni
Step by Step Solution
3.35 Rating (167 Votes )
There are 3 Steps involved in it
a H 0 COH H H HOH bHCCCCCHc ... View full answer

Get step-by-step solutions from verified subject matter experts
Document Format (1 attachment)
15-C-O-O-C (18).docx
120 KBs Word File


