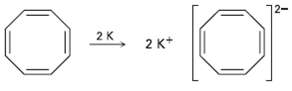
Cyclooctatetraene readily reacts with potassium metal to form the stable Cyclooctatetraene dianion, C 8 H 8 2
Question:
Cyclooctatetraene readily reacts with potassium metal to form the stable Cyclooctatetraene dianion, C8H82?1. Why do you suppose this reaction occurs so easily? What geometry do you expect for the Cyclooctatetraene dianion?
Transcribed Image Text:
2K 2 K* 2.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (12 reviews)
When Cyclooctatetraene accepts two ...View the full answer

Answered By

PALASH JHANWAR
I am a Chartered Accountant with AIR 45 in CA - IPCC. I am a Merit Holder ( B.Com ). The following is my educational details.
PLEASE ACCESS MY RESUME FROM THE FOLLOWING LINK: https://drive.google.com/file/d/1hYR1uch-ff6MRC_cDB07K6VqY9kQ3SFL/view?usp=sharing
3.80+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Why do you suppose that forensic DNA analysis relies principally on short tandem repeats (repeat polymorphisms), rather than single- nucleotide polymorphisms, such as that described on page 669 and...
-
Why do you suppose that large, well-known companies such as Apple, Starbucks, and Facebook prefer to have their shares traded on the NASDAQ rather than on one of the major listed exchanges, such as...
-
Why do you suppose that m x n machine-scheduling algorithms are not widely used in practice? Should optimal rules be more widely used?
-
Fewer than 20% of M&M candies are green. The hypothesis test results in a P-value of 0.0721. Assume a significance level of = 0.05 a. State a conclusion about the null hypothesis. b. Without using...
-
Who is affected by a hospital's decision not to hire smokers? Discuss whether this decision achieves the greatest good for the greatest number of individuals.
-
You are performing CPR on an adult in a confined and crowded space, such as a small restaurant. The environment is noisy, chaotic, and bystanders are not medically trained. Given these challenging...
-
For the HiTech bond of Example 17.4, suppose that default is recognized only at maturity and that no restitution is made, that is, the default is ignored. What is the value of that bond? - Example...
-
Valentine Investigations has the following information for its cash account: Balance, 1/31 ............... $ 7,444 Deposits during February ........... 106,780 Checks written during February ...........
-
1. If you put $1,000 in a savings account at the end of each year for 11 years, how much money will be in the account at the end of the 11th year? Assume that the account earns 11% per annum and...
-
For each of the following four networks, discuss the consequences if a connection fails. a. Five devices arranged in a mesh topology b. Five devices arranged in a star topology (not counting the hub)...
-
Draw the five resonance structures of the cyclopentadienyl anion. Are all carbon carbon bonds equivalent? How many absorption lines would you expect to see in the 1H NMR and 13C NMR spectra of the...
-
Draw an orbital picture of furan to show how the molecule isaromatic. Furan :O:
-
A closed box with a square base is to have a volume of 250 cubic meters. The material for the top and bottom of the box costs $2 per square meter, and the material for the sides costs $1 per square...
-
The main risk for an investor with a negative duration gap is: Group of answer choices Market price risk. Lower interest rates. Higher interest rates.
-
In the Single Index Model (SIM), how is the unsystematic risk of a security measured? Question 18Answer a. By the beta coefficient b. By the R-squared value c. By the residual return d. By the...
-
Consider a 6% T-note with 1.5 years to maturity. Spot rates (expressed as semiannual yields to maturity) are: 6 months = 5%, 1 year = 6% and 1.5 years = 7%. If the note is selling for $992, compute...
-
If the Fed decreases interest rates, this should cause a capital outflow and the value of the USD to Group of answer choices appreciate depreciate remain unchanged none of the above
-
An investor has a position of $1,000,000 in shares of an investment bank called Black Plainly (BP). BP's yearly volatility is 20% and yearly expected returns is 10%. What is our investor's Value at...
-
Prove the first part of Theorem 3.7. That is let \(\left\{X_{n}ight\}_{n=1}^{\infty}\) be a sequence of random variables, \(c\) be a real constant, and \(g\) be a Borel function on \(\mathbb{R}\)...
-
Use this circle graph to answer following Exercises. 1. What fraction of areas maintained by the National Park Service are designated as National Recreation Areas? 2. What fraction of areas...
-
For the reaction, A(g) 2 B( g), K c = 4.0. A reaction mixture at equilibrium contains [A] = 1.0 M. What is the concentration of B in the reaction mixture? (a) 0.50 M (b) 1.0 M (c) 2.0 M (d) 4.0 M
-
(a) Show that D-glucose, D-mannose, and D-fructose all give the same osazone. Show the structure and stereochemistry of this osazone. (b) D-Talose is an aldohexose that gives the same osazone as...
-
Show that Ruff degradation of D-mannose gives the same aldopentose (D-arabinose) as does D-glucose.
-
D-Lyxose is formed by Ruff degradation of galactose. Give the structure of D-lyxose. Ruff degradation of D-lyxose gives D-threose. Give the structure of D-threose.
-
Sunland Corp. designs and builds custom harvesting equipment for logging companies across Canada. The company, which is publicly traded, has a May 31 year end. On February 18, 2024, Sunland signed a...
-
Kubin Company's relevant range of production is 18,000 to 22,000 units. When it produces and sells 20,000 units, its average costs per unit are as follows: Average Cost per Unit Fixed administrative...
-
The following balance sheet for the Los Gatos Corporation was prepared by a recently hired accountant. In reviewing the statement you notice several errors. LOS GATOS CORPORATION Balance Sheet At...

Study smarter with the SolutionInn App


