Question: Determine by the Kremser method the separation that can be achieved for the absorption operation indicated in Figure for the following combinations of conditions: (a)
Determine by the Kremser method the separation that can be achieved for the absorption operation indicated in Figure for the following combinations of conditions:
(a) Six equilibrium stages and 75 psia operating pressure,
(b) Three equilibrium stages and 150 psia operating pressure,
(c) Six equilibrium stages and 150 psia operating pressure. At 90°F and 75 psia, the K-value of nC10 =0.0011.
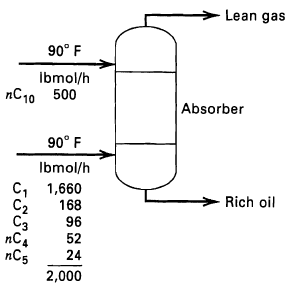
Lean gas 90 F Ibmol/h 10 500 Absorber 90 F Ibmol/h C, 1,660 C2 C nC5 Rich oil 168 96 52 24 2,000
Step by Step Solution
3.34 Rating (157 Votes )
There are 3 Steps involved in it
Use the Kremser method with Eqs 548 for fraction not absorbed A 550 for fr... View full answer

Get step-by-step solutions from verified subject matter experts
Document Format (1 attachment)
37-E-C-E-S-P (218).docx
120 KBs Word File


