Determine the carburizing time necessary to achieve a carbon concentration of 0.45 wt% at a position 2
Question:
Determine the carburizing time necessary to achieve a carbon concentration of 0.45 wt% at a position 2 mm into an iron?carbon alloy that initially contains 0.20 wt% C. The surface concentration is to be maintained at 1.30 wt% C, and the treatment is to be conducted at 1000?C. Use the diffusion data for g-Fe in Table 5.2.
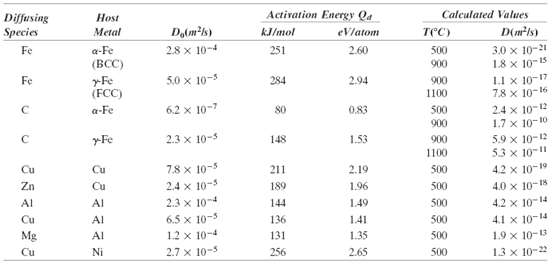
Transcribed Image Text:
Diffusing Activation Energy Qa Host Calculated Values Species Do(m²ls) Metal kJ/mol eVlatom D(mls) тес) Fe 2.8 x 10-4 a-Fe 251 2.60 500 3.0 x 10-21 18 x 10-15 LI x 10-17 78 x 10-16 (BCC) 900 Fe rFe (FCC) 5.0 x 10- 284 2,94 900 1100 a-Fe 6.2 x 10-7 80 0.83 24 x 10-12 1.7 x 10-10 50 900 2.3 x 10-5 rFe 1.53 148 900 5.9 x 10-12 53 x 10-11 1100 Cu 7.8 x 10-5 2.4 x 10-5 Cu 211 2.19 500 4.2 x 10-19 Zn Cu 189 1.96 500 4.0 x 10-18 Al 2.3 x 104 Al 144 1.49 500 4.2 x 10-14 Cu 6.5 x 10-3 12 x 10-4 2.7 x 10-5 Al 136 141 500 4.1 x 10-14 Mg Al 131 1.35 500 1.9 x 10-3 Cu Ni 256 2.65 500 1.3 x 10-2
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 58% (12 reviews)
In order to solve this problem it is first necessary ...View the full answer

Answered By

OTIENO OBADO
I have a vast experience in teaching, mentoring and tutoring. I handle student concerns diligently and my academic background is undeniably aesthetic
4.30+
3+ Reviews
10+ Question Solved
Related Book For 

Fundamentals of Materials Science and Engineering An Integrated Approach
ISBN: 978-1118061602
4th Edition
Authors: David G. Rethwisch
Question Posted:
Students also viewed these Materials Science Engineering questions
-
Determine the carburizing time necessary to achieve a carbon concentration of 0.30 wt% at a position 4 mm into an iron-carbon alloy that initially contains 0.10 wt% C. The surface concentration is to...
-
The Space time necessary to achieve 70 percent conversion in a CSTR is 3h. Determine the reactor volume required to process 4 cubic feet / min. What is the space velocity for this system?
-
An alloy of iron and carbon was treated with sulfuric acid, in which only iron reacts. 2Fe(s) + 3H2SO4(aq) Fe2(SO4)3(aq) + 3H2(g) If a sample of alloy weighing 2.358 g gave 0.1152 g of hydrogen,...
-
Goddard Company has used the FIFO method of inventory valuation since it began operations in 2015. Goddard decided to change to the average cost method for determining inventory costs at the...
-
Distinguish intentional torts from negligent torts?
-
Refer to all of the facts in Problem 12-6. Required 1. Using the format in the chapter's appendix, prepare a statement of cash flows work sheet. 2. Prepare a statement of cash flows for 2012 using...
-
Explain the relationship between TPSs and FBSs. A functional business system (FBS) is an information system designed to improve the efficiency and performance of a specific functional area within an...
-
Towerton Financial Services13 Towerton Financial Services, a brokerage firm, started with a focus on stock trading and mutual funds. As the business grew, Towerton diversified into two new product...
-
S On June 30, 2024, Georgia-Atlantic, Incorporated leased warehouse equipment from Builders, Incorporated The lease agreement calls for Georgia-Atlantic to make semiannual lease payments of $681,881...
-
India has an import-competing auto sector that operates under perfectly competitive conditions. Currently, there are no restrictions on auto imports, or any taxes or subsidies. Auto production in...
-
Show that is also a solution to Equation 5.4b. The parameter B is a constant, being independent of both x and t. C, VDE -exp Dt 4 Dt
-
An FCC iron-carbon alloy initially containing 0.35 wt% C is exposed to an oxygen-rich and virtually carbon-free atmosphere at 1400 K (1127C). Under these circumstances the carbon diffuses from the...
-
Indicate how the following business transactions affect the basic accounting equation. (a) Paid cash for janitorial services. (b) Purchased equipment for cash. (c) Received cash in exchange for...
-
What are the steps in writing a report?
-
For written reports, what is the difference between the factual format and the administrative format?
-
What is TARP and how was it funded? What is meant by the term lender of last resort and how does it relate to the financial crisis of 20072008? How do government and Federal Reserve emergency loans...
-
What are the different techniques or methods for identifying candidate solutions? If you had to choose just one of these methods, which would it be and why?
-
What are some of the benefits of prototyping?
-
The data file collegetown contains data on 500 single-family houses sold in Baton Rouge, Louisiana, during 2009-2013. The data include sale price in \(\$ 1000\) units, PRICE, and total interior area...
-
Maria Castigliani is head of the purchasing department of Ambrosiana Merceti, a medium-sized construction company. One morning she walked into the office and said, The main problem in this office is...
-
Calculate the emf x of a dry cell for which a potentiometer is balanced when R x = 1.200 , while an alkaline standard cell with an emf of 1.600 V requires R s = 1.247 to balance the potentiometer.
-
Is it possible to produce an iron-carbon alloy of eutectoid composition that has a minimum hardness of 90 HRB and a minimum ductility of 35%RA? If so, describe the continuous cooling heat treatment...
-
Is it possible to produce an iron-carbon alloy that has a minimum tensile strength of 690 MPa (100,000 psi) and a minimum ductility of 40%RA? If so, what will be its composition and microstructure...
-
It is desired to produce an iron-carbon alloy that has a minimum hardness of 175 HB and a minimum ductility of 52%RA. Is such an alloy possible? If so, what will be its composition and microstructure...
-
We review long-lived assets for impairment when circumstances indicate the carrying amount of an asset may not be recoverable based on the undiscounted future cash flows. If the carrying amount of...
-
Hal's Heavenly Creations offers its employees the option of contributing up to 6% of their salaries to a voluntary retirement plan, with the employer matching their contribution. The company also...
-
Altira Corporation provides the following information related to its inventory during the month of August 2024: August 1 Inventory on hand-2,300 units; cost $6.40 each. August 8 Purchased 11,500...

Study smarter with the SolutionInn App


