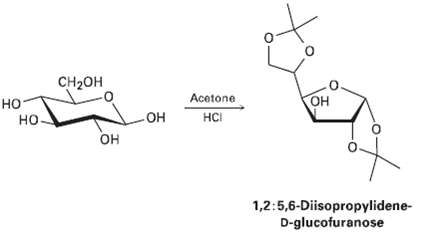
D-Glucose reacts with acetone in the presence of acid to yield the non-reducing 1, 2: 5, 6-diisopropylidenc-D-glucofuranose.
Question:
D-Glucose reacts with acetone in the presence of acid to yield the non-reducing 1, 2: 5, 6-diisopropylidenc-D-glucofuranose. Propose amechanism.
Transcribed Image Text:
Cн2он Acetone Он но HCI HO но- он 1,2:5,6-Diisopropylidene- D-glucofuranose it
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 83% (6 reviews)
Glucopyranose is in equilibrium with glucofuranose Reaction with t...View the full answer

Answered By

Aysha Ali
my name is ayesha ali. i have done my matriculation in science topics with a+ . then i got admission in the field of computer science and technology in punjab college, lahore. i have passed my final examination of college with a+ also. after that, i got admission in the biggest university of pakistan which is university of the punjab. i am studying business and information technology in my university. i always stand first in my class. i am very brilliant client. my experts always appreciate my work. my projects are very popular in my university because i always complete my work with extreme devotion. i have a great knowledge about all major science topics. science topics always remain my favorite topics. i am also a home expert. i teach many clients at my home ranging from pre-school level to university level. my clients always show excellent result. i am expert in writing essays, reports, speeches, researches and all type of projects. i also have a vast knowledge about business, marketing, cost accounting and finance. i am also expert in making presentations on powerpoint and microsoft word. if you need any sort of help in any topic, please dont hesitate to consult with me. i will provide you the best work at a very reasonable price. i am quality oriented and i have 5 year experience in the following field.
matriculation in science topics; inter in computer science; bachelors in business and information technology
_embed src=http://www.clocklink.com/clocks/0018-orange.swf?timezone=usa_albany& width=200 height=200 wmode=transparent type=application/x-shockwave-flash_
4.40+
11+ Reviews
14+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Glucose reacts with ammonia in the presence of a trace of acid to give predominantly -D- glucopyranosylamine. Propose a reasonable mechanism for this transformation. Why is only the hydroxy group at...
-
In a sulfuric acid plant, sulfur is burned in the presence of excess oxygen to produce sulfur dioxide, (SO,) which in turn is further reacted in the next step with oxygen in a converter to produce...
-
In the presence of light, chlorine can substitute for one (or more) of the hydrogens in an alkane. For the following reactions, draw the possible monochlorination products. hr 2,2-dimethylpropane Cl2...
-
Why does Sam Goldman go out of his way to talk about the importance of d.lights business plan? In what ways do you think having a meticulously crafted business plan helped d.light in its launch...
-
What are some questions to ask when gauging the audience's needs during the planning of a persuasive message?
-
Consider a metal can placed coaxially inside a long solenoid. The metal can be modelled as a thin cylindrical shell of radius r. The magnetic field due to the solenoid varies with time t as kt where...
-
Verify the alternative formulas for computing \(S S T\) and \(S S(T r)\) given on page 399 . Data From Page 399 k ni SST=-C i=1 j=1 k SS(Tr) = ni i=1 where C, called the correction term for the mean,...
-
What types of mechanisms can be used to help increase collaboration across business and IS leaders?
-
Huegel Hollow Resort has ordered 24 rotomolded kayaks from Current Designs. Each kayak will be formed in the rotomolded oven, cooled, and then have the excess plastic trimmed away. Then, the hatches,...
-
You are given the following payoff table (in units of thousands of dollars) for a decision analysis problem: (a) Which alternative should be chosen under the maximin payoff criterion? (b) Which...
-
Iso trehalose and neotrehalose are chemically similar to trehalose except that neotrehalose is hydrolyzed only by -glycosidase enzymes, whereas iso trehalose is hydrolyzed by both -and-glycosidase...
-
D-Mannose reacts with acetone to give a diisopropylidene derivative that is still reducing toward Tollens reagent. Propose a likely structure for this derivative.
-
How an auditor of a bank is appointed?
-
1. Tywin Lannister is the sole owner of his family's homeplace, Casterly Rock, comprised of 21 acres. His county tax bill shows that Casterly Rock is appraised at $200,000 (based on a county...
-
Based on you own knowledge of business, which of these scenarios do you think are likely? Are there other implications for the hr functions and hr strategies that you can identify
-
A business may lease office equipment and then make regular payments throughout the term of the lease. At the end of the lease, the business may have the option to purchase the equipment for a...
-
1.1. Find all values of a EC for which the given vector b C is a linear combi- nation of v, v and us, where bm V2 H V = Va 1.2. Let q, Pi. Pa and pa be the polynomials in P,(C) defined by q(2)...
-
7) For a patients consent to be valid, several criteria will need to be met. Briefly describe two requirements of obtaining valid consent. 8) Explain the difference between Implied Consent and...
-
A small company wishes to setup a fund that can be used for technology purchases over thenext6years. Their forecast is for \($12\),000 to be needed at the end of year 1, decreasing by \($2\),000 each...
-
In Problem use geometric formulas to find the unsigned area between the graph of y = f(x) and the x axis over the indicated interval. f(x) = x + 5; [0, 4]
-
A mixture of NaCN and NaHSO 4 consists of a total of 0.60 mol. When the mixture is dissolved in 1.0 L of water and comes to equilibrium, the pH is found to be 9.9. Find the amount of NaCN in the...
-
Using the peroxy acid epoxidation of an alkene and the ring opening of an epoxide, devise a two-step synthesis of 1, 2-butanediol from 1-butene.
-
Write an equation for the reaction of ethylene oxide with a. 1 mole of HCl b. Excess HCl c. Phenol + H+ d. Phenylmagnesium bromide
-
CH3CH2OCH2CH2OH (ethyl cellosolve) and CH3CH2OCH2CH2OCH2CH2OH (ethyl carbitol) are solvents used in the formulation of lacquers. They are produced commercially from ethylene oxide and certain other...
-
Explain the concept of hybrid thread models in operating systems. How do they attempt to combine the benefits of user-space and kernel-space threading ?
-
Describe the characteristics of the six stages of development for intimacy in a romantic relationship using a real or hypothetical example to illustrate the general developmental path.
-
Consider the following payoff matrix: Player 1 Strategy B Strategy A Strategy A (4,3) (6,2) Player 2 Strategy B (3,5) (2,4) Is the payoff matrix symmetric (Yes or No)?

Study smarter with the SolutionInn App


