Draw the following molecules in chair conformations, and tell whether the ring sub-stituents are axial orequatorial: CH
Question:
Draw the following molecules in chair conformations, and tell whether the ring sub-stituents are axial orequatorial:
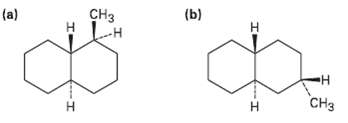
Transcribed Image Text:
CHз (b) (a) н н CHз Н н
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 90% (20 reviews)
Both ring systems are transfused and both hydro...View the full answer

Answered By

Jacob Festus
I am a professional Statistician and Project Research writer. I am looking forward to getting mostly statistical work including data management that is analysis, data entry using all the statistical software’s such as R Gui, R Studio, SPSS, STATA, and excel. I also have excellent knowledge of research and essay writing. I have previously worked in other Freelancing sites such as Uvocorp, Essay shark, Bluecorp and finally, decided to join the solution inn team to continue with my explicit work of helping dear clients and students achieve their Academic dreams. I deliver, quality and exceptional projects on time and capable of working under high pressure.
4.90+
1252+ Reviews
2844+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Using Newman projections, draw each of the following molecules in its most stable conformation with respect to the bond indicated: (a) 2-methylbutane, C2-C3 bond; (b) 2,2-dimethylbutane, C2-C3 bond;...
-
Draw the two chair conformations of the sugar α-(+)-glucopyranose, one form of the sugar glucose. Which of these two forms is the major one at equilibrium? Explain. CH2OH OH ...
-
Referring to Table 10.3, arrange the following molecules in order of increasing dipole moment: H2O, H2S, H2Te, H2Se?
-
Consider the exchange rate between South Korea and Costa Rica. Typically, exchange rates vary over time, sometimes quite dramatically. The scenarios present various changes that may affect the...
-
Write four messages of no more than 140 characters each (short enough to work as Twitter tweets, in other words) to persuade other college students to take the business communication course. Think of...
-
A light bulb glows because it has resistance. The brightness of a light bulb increases with the electrical power dissipated in the bulb. (a) In the circuit shown in Fig. Q25.14a, the two bulbs A and...
-
Consider the multiple regression model fit to the gasoline mileage data in Problem 3.5. Problem 3.5 Consider the gasoline mileage data in Table B.3. a. Construct a normal probability plot of the...
-
Price Company issues $400,000, 20-year, 7% bonds at 101. Prepare the journal entry to record the sale of these bonds on June 1, 2017.
-
Problem: Module 3 Textbook Problem 5 Learning Objective: 3-6 Using the straight-line method show how bonds issued at a discount. affect financial statements Diaz Company issued $91,000 face value of...
-
Ellipses Corp is a small business that operates in Herndon, VA. The company is located at10 Period Lane, Herndon, VA 20170. Its federal Employer Identification Number is 77-7777777, and its...
-
Propose mechanistic pathways for the biosynthetic formation of the followingterpenes: (b) (a) a-Pinene y-Bisabolene
-
Lithocholic acid is an A-B cis steroid found in human bile. Draw Lithocholic acid showing chair conformations as in figure, and tell whether the hydroxyl group at C3 is axialequatorial. An A-B trans...
-
Catalytic hydrogenation of 1,4-dimethylcyclopentene yields a mixture of two products. Identify them. One of them is formed in much greater amounts than the other (observed ratio =10:1). Which one is...
-
'Borrower') and Lighthouse Bank plc (the 'Lender') THIS DEED is dated 1 March 2023. 1. DEFINITIONS AND INTERPRETATION 1.1 The definitions and rules of interpretation in this clause apply in this...
-
You are an intern in the corporate office of MicRonalds Pty Ltd which runs fast food outlets in different cities around Australia. You have received instructions from Kylie Eagle, the company's...
-
Harrison and Gloria are shareholders of a Canadian-Controlled Private Corporation (CCPC). In the past year, the CCPC received a death benefit of $300,000 from a life insurance policy which had an...
-
A nice and concise chapter. I enjoy how it lays out a framework for attacking contractual issues: First, is tthere even a "contract" ( consideration)? ; Second, is the contract even legal ? Third,...
-
The Statement of Work is changing but the client does not want to take all the risk. What would be the best contract type for the client?
-
For each case below, plot the Nichols chart and closed-loop frequency response. If the system is stable find the gain margin, phase margin, peak closed-loop frequency response, and system bandwidth....
-
Use a calculator to evaluate the expression. Round your result to the nearest thousandth. V (32 + #)
-
Determine the percent ionization of a 0.225 M solution of benzoic acid.
-
Write equations for the reactions of furan with a. Br2 b. HNO3 c. CH3COCl (acetyl chloride), SnCl4
-
Write equations for the reactions 2-methylfuran with the reagents shown in Problem 13.16. In Problem 13.16 Write an equation for each of the following reactions: a. Quinoline (page 395) + HCl b....
-
Although electrophilic substitution occurs at C2 in pyrrole, it occurs predominantly at C3 in indole. Suggest an explanation.
-
What theorists are most likely to argue the city's form and growth come from decisions made by people and organizations that control wealth and other key resources?
-
Explain the difference between glucogenic and ketogenic amino acids
-
Describe what is an arrangement where workers who don't join a union must make payments equal to union dues and fees to get union representation services?

Study smarter with the SolutionInn App


