Draw the two chair conformations of menthol, and tell which is morestable. C Menthol CH(CH3)2
Question:
Draw the two chair conformations of menthol, and tell which is morestable.
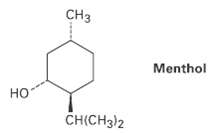
Transcribed Image Text:
Cнз Menthol но CH(CH3)2
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 58% (12 reviews)
From the flatring drawing you can see that the methyl group and the OH group have a cis relation...View the full answer

Answered By

Carly Cimino
As a tutor, my focus is to help communicate and break down difficult concepts in a way that allows students greater accessibility and comprehension to their course material. I love helping others develop a sense of personal confidence and curiosity, and I'm looking forward to the chance to interact and work with you professionally and better your academic grades.
4.30+
12+ Reviews
21+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Draw the two chair conformations of the sugar α-(+)-glucopyranose, one form of the sugar glucose. Which of these two forms is the major one at equilibrium? Explain. CH2OH OH ...
-
Draw the two chair conformations of cis-1-chloro-2-methylcyclohexane. Which is more stable, and by how much?
-
Draw the two chair conformations of trans-1-chloro-2-mcthylcyclohexane. Which is more stable?
-
A strange function. Consider McCarthys 91 function: public static int mcCarthy(int n) { if (n > 100) return n - 10; return mcCarthy(mcCarthy(n+11)); } Determine the value of mcCarthy(50) without...
-
Tammy Jackson purchased 100 shares of All-American Manufacturing Company stock at $31.50 a share. One year later, she sold the stock for $38 a share. She paid her broker a $15 commission when she...
-
Suppose that the equation yt = a + (t + (1xt1 + ...+(kxtk + u, satisfies the sequential exogeneity assumption in equation (11.40). (i) Suppose you difference the equation to obtain (yt = ( + (1 (xt1...
-
A uniaxial off-axis tensile test is conducted as shown in Figure 4.5. Using the Tsai-Hill criterion and assuming that the lamina strengths are the same in tension and compression, develop an equation...
-
A pet store currently uses a legacy flat file system to store all of its information. The owner of the store, Peter Corona, wants to implement a Web-enabled database application. This would enable...
-
Choose one of the four basic leader behavior styles suggested by Hersey and Blanchard. Discuss the specifics of the task and relationship behaviors of the chosen style. Provide a realistic example of...
-
A yellow pine rod (SG = 0.65) is 5 cm by 5 cm by 2.2 m long. How much lead (SG = 11.4) is needed at one end so that the rod will float vertically with 30 cm out of the water? 0.3 m Wood 1.9 m Lead
-
Galactose, a sugar related to glucose, contains a six-membered ring in which all the substituents except the ?OH group indicated below in red are equatorial. Draw galactose in its more stable chair...
-
There are four cistransisomers of menthol (Problem 4.37), including the one shown. Draw the other three.
-
Why do some believe that one should think regional and downplay global? Do you agree? Why?
-
knowing why : what factors have motivated you to choose this career pathway? These should include: occupational interests - strengths - values - knowing what : what career are you interested in...
-
Shuggy Otis, an executive at Slapfish Corp. (SC) intends to retire in 11 years. SC just announced that it will start depositing $500.00 at the end of each quarter into each of its workers' retirement...
-
Icebreaker needs to gather insights about people in New Zealand who are parents of children aged 0 to 4 years, or soon-to-be parents, who would be interested in buying the new apparel range made from...
-
The value of total assets in Valera inc company equals to 100,000. The balance sheet of the company documents the following values: fixed assets of 50,000, long term liabilities of 20,000, current...
-
This year, Company ABC purchases Class 8 (20%) Assets for $20,000, what is the maximum CCA for this year?
-
A metaphor places two dissimilar objects into a close relationship such that A is B, whereas a simile compares two objects such that A is like B. A and B, however dissimilar, share some quality that...
-
Discuss whether responsible human resources management should apply different standards for the home company and suppliers, for developed countries and developing countries, and for large companies...
-
Arrange the following substances in the order in which you would expect their boiling points to increase: CCl 4 , Cl 2 , ClNO, N 2 .
-
Provide a mechanism for the following reaction, based on your knowledge of the reaction of esters with Grignard reagents. OH MgBr (2 equiv.) Cl (2) NH,CI
-
Provide retrosynthetic analyses and syntheses for each of the following alcohols, starting with appropriate alkyl or aryl halides. (a) (b) (c) (d) (e) (f) OH (three ways) OH (three ways) (two ways)...
-
Consider the allylic bromination of cyclohexene labeled at C3 with 13C. Neglecting stereoisomers, what products would you expect from this reaction? NBS, ROOR heat (-13C-labeled position)
-
A scuba diver is exploring a shipwreck at a depth of 30 meters in the ocean. The diver's tank contains 5.0 moles of compressed air at a temperature of 25C. What is the pressure of the air in the tank...
-
1. Determine the atmospheric pressure at a location where the barometric reading is 720 mm Hg. 2. A pressure gauge connected to a tank reads 52 psi at a location where the barometric reading is 29.6...
-
Assume that a bond has an annual coupon rate of 6.80 percent, but makes coupon payments on a semi-annual basis, has a par value of $1,000, a yield to maturity of 7.20 percent, and a current price of...

Study smarter with the SolutionInn App


